Specific Heat at Constant Volume (Cv)
The rate of change of internal energy with respect to absolute temperature at constant volume is known as specific heat at constant volume (Cv).
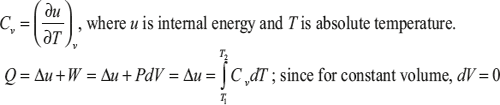
Enthalpy is sum of internal energy and product of pressure and volume, i.e., h = u + PV. But
Q = ∂u + PdV = ∂u + ∂(PV) = ∂(u + PV) = ∂h
since dP = 0 at constant pressure.
Specific Heat at Constant Pressure (Cρ)
The rate of change of enthalpy with respect to absolute temperature when pressure is constant is known as specific heat at constant pressure (Cp).

Example 1.1: The property of a substance is given as

where u is the specific internal energy (kJ/kg), t is temperature in °C, p is pressure in kN/m2, and v is specific volume (m3/k).
Find the Cv and Cp of the substance.
Solution:

Relationship Between Cρ and Cv
Specific heat capacity of a gas is the amount of heat required to raise the temperature by 1°C of unit mass of the gas. We will use here specific values of the state variables (of the variable divided by the mass of the substance). The value of the constant is different for different materials and depends on the process. It is not a state variable.
If we are considering a gas, it is most convenient to use forms of the thermodynamics equations based on the enthalpy of the gas. From the definition of enthalpy:
h = u + pv
where h is the specific enthalpy, p is the pressure, v is the specific volume, and u is the specific internal energy. During a process, the values of these variables change. Let us denote the change by Δ. For a constant pressure process the enthalpy equation becomes
Δh = Δu + pΔv
The enthalpy, internal energy, and volume are all changed, but the pressure remains the same. From our derivation of the enthalpy equation, the change of specific enthalpy is equal to the heat transfer for a constant pressure process:
Δh = cpΔT
where ΔT is the change of temperature of the gas during the process, and c is the specific heat capacity. We have added a subscript p to the specific heat capacity at a constant pressure process.
The equation of state of a gas relates the temperature, pressure, and volume through a gas constant R. The gas constant is derived from the universal gas constant, but has a unique value for every gas.
pv = RT
For a constant pressure process:
pΔv = RΔT
Now let us consider a constant volume process with a gas that produces exactly the same temperature change as the constant pressure process that we have been discussing. Then the first law of thermodynamics tells us:
Δu = Δq − Δw
where q is the specific heat transfer and w is the work done by the gas. For a constant volume process, the work is equal to zero. And we can express the heat transfer as a constant times the change in temperature. This gives
Δu = cv ΔT
where ΔT is the change of temperature of the gas during the process and c is the specific heat capacity. We have added a subscript v to the specific heat capacity at a constant volume process. Even though the temperature change is the same for this process and the constant pressure process, the value of the specific heat capacity is different.
Because we have selected the constant volume process to give the same change in temperature as our constant pressure process, we can substitute the expression given above for Δu into the enthalpy equation. In general, we cannot make this substitution because a constant pressure process and a constant volume process produce different changes in temperature. If we substitute the expressions for Δu, pΔv, and Δh into the enthalpy equation we obtain:
cpΔT = cvΔT + RΔT
Dividing the above equation by ΔT, we get
cp = cv + R
The specific heat constants for constant pressure and constant volume processes are related to the gas constant for a given gas. This rather remarkable result has been derived from thermodynamic relations, which are based on observations of physical systems and processes. Using the kinetic theory of gases, this same result can be derived from considerations of the conservation of energy at a molecular level.
We can define an additional variable called the specific heat ratio, which is given the Greek symbol γ (Gamma), which is equal to cp divided by cv:
γ = cp/cv
γ is a number whose value depends on the state of the gas. For air, γ = 1.4 for standard conditions.
For monoatomic gas (γ) = Cp/Cv = 5/3 = 1.66
For diatomic gas (γ) = Cp/Cv = 7/5 = 1.40
For triatomic gas (γ) = Cp/Cv = 8/6 = 1.33

Leave a Reply