It is not always useful to know only how much heat energy is needed to raise the temperature of a body. For instance, if you are boiling a kettle, you are more interested in how long it will be before you can make the tea. The quantity of heat energy needed has to be related to the power available, in this case the rating of the heating element of the kettle, and if you have a typical kettle of, say, 2 kW, it means that in 1 second it provides 2000 joules of heat energy.
Remember that power is the rate at which the energy is delivered, i.e. work, or heat energy delivered, divided by time taken.
Let us say the kettle contains 2 kg of water and is at a room temperature of 18°C, and the kettle is 2 kW. Specific heat of water = 4.2 kJ/kgK.
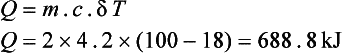
This is the heat energy required to raise the temperature of the water to 100°C. The kettle is producing 2 kW, i.e. 2 kJ/s. Therefore,
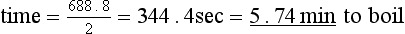
The specific heats of gases
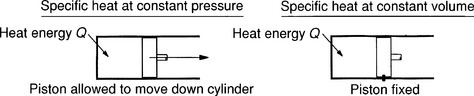
Solids have a value of specific heat which varies only slightly with temperature. On the other hand, gases can have many different values of specific heat depending on what happens to it while it is being heated or cooled. The two values which are used are the specific heat at constant pressure, cp, and the specific heat at constant volume, cv. See Figure 2.1.1.

Leave a Reply