The expansion and compression of gases, such as air and combustion gases, is a very important subject in the study of the operation of compressors, all types of reciprocating engines, gas turbines, and in pneumatic systems.
We need to be able to predict how the volume, temperature and pressure of a gas inter-relate in a process in order to design systems in which gas is the working medium. The cycle of operations of your car engine is a good example of the practical application of this study.
In this chapter, therefore, we look at the different ways in which a gas can be expanded and compressed, what defines the ‘system’, the significance of reversibility and the First Law of Thermodynamics. In order to be able to calculate changes in properties during a process, gas law expressions and equations are introduced.
Boyle’s and Charles’ laws
The gases we deal with are assumed to be ‘perfect gases’, i.e. theoretically ideal gases which strictly follow Boyle’s and Charles’ laws. What are these laws?
Boyle’s law. This says that if the temperature of a gas is kept constant, the product of its pressure and its volume will always be the same. Hence,

or,

This means that if you have a quantity of gas and you change its pressure and therefore its volume, as long as the temperature is kept constant (this would require heating or cooling), you will always get the same answer if you multiply the pressure by the volume.
Charles’ law. This says that if you keep the pressure of a gas constant, the value of its volume divided by its temperature will always be the same. Hence,
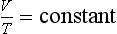
or,

These laws can be remembered separately, but from an engineer’s point of view they are better combined to give a single very useful expression. This is,
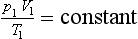
or,

It is important to remember that this expression is always valid, no matter what the process, and when doing calculations it should always be the first consideration.
• There are some instances, as we shall see later, where the pressure must be in N/m2, but for the time being, bar can be used. 1 bar = 105 N/m2.
• Temperature is always absolute, i.e. K. The best practice is to convert all temperatures to K immediately by adding 273 to your centigrade temperatures. There is an exception to this when you have a temperature difference, since this gives the same value in °C or K.
Units
T = absolute temperature (K) (°C + 273)
Example 2.2.1
A perfect gas in an engine cylinder at the start of compression has a volume of 0.01 m3, a temperature of 20°C and a pressure of 2 bar.
The piston rises to compress the gas, and at top dead centre the volume is 0.004 m3 and the pressure is 15 bar. Find the temperature. See Figure 2.2.1.
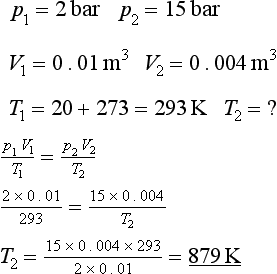
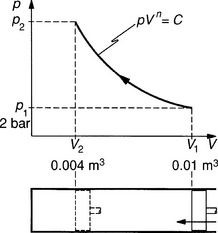
Figure 2.2.1 p/V diagram. Example 2.2.1
The geometry of the cylinder and piston is fixed, so the only way in which the value of temperature (or pressure) at the end of compression can be different for the same initial conditions is if there are different rates of heat energy transfer across the cylinder walls, i.e. how much cooling there is of the engine.
The pressure/volume (p/V) diagram
It is important to examine the pressure/volume diagram for a process, because this gives a visual appreciation of what is happening, and also, some formulae are derived from the diagrams. p/V diagrams of actual engine cycles are used to calculate power and to adjust timing.
The p/V diagram for Example 2.2.1 is shown in Figure 2.2.1. The volume axis is drawn vertically. To aid understanding, the piston and cylinder are shown beneath the diagram in this case. The direction of the arrow indicates if the process is an expansion or a compression, and the curve would normally be labelled to show the type of process occurring.
Reversibility
We draw the p/V diagrams for our processes assuming that the processes are reversible. Put simply, this means that the process can be reversed so that no evidence exists that the process happened in the first place. The best analogy is of a frictionless pendulum swinging back and forth without loss of height.
However, this is not what happens in practice, because of temperature differences, pressure differences, and turbulence within the fluid during the process, and because of friction, all of which are ‘irreversibilities’.
The only way in which a reversible process could be achieved is to allow equilibrium to be reached after each of an infinite number of stages during the process, i.e. extremely slowly to let things settle down. This, unfortunately, would take an infinite time.
Strictly speaking, the p/V diagrams should be dotted to show that we are dealing with irreversible processes, but for convenience solid lines are usually used. We assume that at the end states our gas is in equilibrium.
The significance of the concept of reversibility becomes apparent in later studies of thermodynamics.

Leave a Reply