Previously, to define electricity, several theories were developed through experiments and by observing its behaviour. Among them, the well-explained theory was ‘Modern Electron Theory of Matter’. This theory was developed based on the research works of the famous scientists Sir William Crooks, J. J. Thomson, Robert A. Milliken, Sir Earnest Rutherford, and Neils Bohr.
According to modern electron theory, every matter (whether solid, liquid, or gas) consists of tiny divisible particles called ‘molecules’. A molecule is further made up of very minute particles called ‘atoms’.
Generally, an atom consists of two main parts, namely nucleus and extra nucleus.
- Nucleus: The central part of an atom, which contains protons and neutrons, is called nucleus. A proton has positive charge (1.602 × 10−19 coulombs), whereas a neutron has no charge. Therefore, the nucleus of an atom possesses positive charge. In the nucleus, the protons and neutrons are held together with tremendous force of attraction. The mass of proton is equal to the mass of neutron, and this total mass constitutes the entire mass of an atom.
- Extra nucleus: The outer part of an atom, which contains only electrons, is called ‘extra nucleus’. An electron has negative charge (1.602 × 10−19 coulombs) equal to that of proton. The mass of an electron is nearly 1⁄1,840 times to that of a proton and it is usually neglected. Therefore, the entire mass of an atom is constituted by the nucleus of an atom.
Atomic weight = Number of protons + Number of neutrons in the nucleus
The electrons are not stationary particles; they move around the nucleus in different paths or orbits in a disciplined manner. The shape of an orbit is more or less elliptical, but for simplicity, circular orbits are shown in Figure 1.1. The number of electrons is same as that of protons in an atom. Therefore, an atom on the whole is neutral. The number of protons or electrons in an atom is called atomic number.
Atomic number = Number of protons or electrons in an atom
To know the electrical behaviour of a matter, its simple atomic structure is required to be drawn. To draw simple atomic structure of an atom, the number of electrons in any orbit is determined by the following rules:
- The number of electron in any orbit is given by relation 2n2, where ‘n’ is the number of orbit counting from nucleus and going outwards.For example, First orbit has 2 × 12 = 2 electronsSecond orbit has 2 × 22 = 8 electronsThird orbit has 2 × 32 = 18 electrons and so on.
- The last orbit cannot have more than eight electrons.
- The last but one orbit cannot have more than 18 electrons.
Simple atomic structure of silver (Ag), copper (Cu), and aluminium (Al) are shown in Figure 1.1.
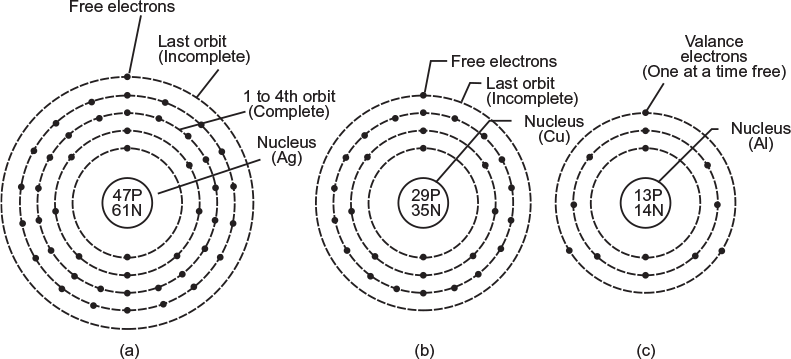
Fig. 1.1 Atomic structure (Bohr’s model) of various metals. (a) Silver (Ag), (b) Copper (Cu) (c) Aluminium (Al)
Figure 1.1(a) shows the atomic structure of silver (Ag), where
Atomic weight of silver = 108
Atomic number of silver = 47
Number of electrons = Number of protons = 47
Number of neutron = Atomic weight − atomic number
Hence, number of neutron = 108 − 47 = 61
Number of electrons in the first orbit = 2 × 12 = 2
Number of electrons in the second orbit = 2 × 22 = 8
Number of electrons in the third orbit = 2 × 32 = 18
Number of electrons in the fourth orbit = 18
Number of electrons in the last orbit = 1
The last orbit of an atom can have maximum eight electrons but silver atom has only one electron in the last (fifth) orbit. Hence, it is an incomplete orbit. This electron of the outermost orbit has poor binding forces with the nucleus and moves freely from one atom to the other at random. Therefore, it is known as ‘free electron’.
Figure 1.1(b) shows the atomic structure of copper. The atomic weight and atomic number of copper are 64 and 29, respectively. The 29 electrons are distributed in the first, second, third, and fourth orbits as 2, 8, 18, and 1, respectively. There is only one electron in the outermost (fourth) orbit, which moves at random from one atom to the other in the matter itself, and is known as free electron.
Figure 1.1(c) shows the atomic structure of aluminium. The atomic weight and atomic number of aluminium are 27 and 13, respectively. The 13 electrons are distributed in first, second, and third orbits as 2, 8, and 3, respectively. The last orbit of the atom can have maximum eight electrons, but the aluminium atom has only three electrons. Hence, it is an incomplete orbit. Of the three electrons of the outermost orbit, only one electron is free to move from one atom to the other and is called free electron.
The following conclusions were drawn from the discussion:
- Every mater is electrical in nature since it contains the charged particles such as electrons and protons.
- Under ordinary conditions, a body is electrically neutral since every atom of the body material is having the same number of protons and electrons.
- Each matter differs from the other since they have different atomic number and structure.
- An atom cannot have more than one free electron at the same instant. For example, in case of aluminium, there are three electrons in the outermost (third) orbit but only one of them is free at a time.
- Silver is more conductive material than the other two (i.e., copper and aluminium), since in case of silver atomic structure, the free electron is in the fifth orbit and is more loosely attached. However, copper is more conductive than aluminium since the free electron in case of copper atomic structure is in the fourth orbit. The aluminium is the poorest conducting material of the three, since in its atomic structure the free electron is in the third orbit and is more rigidly attached to the nucleus.
Leave a Reply