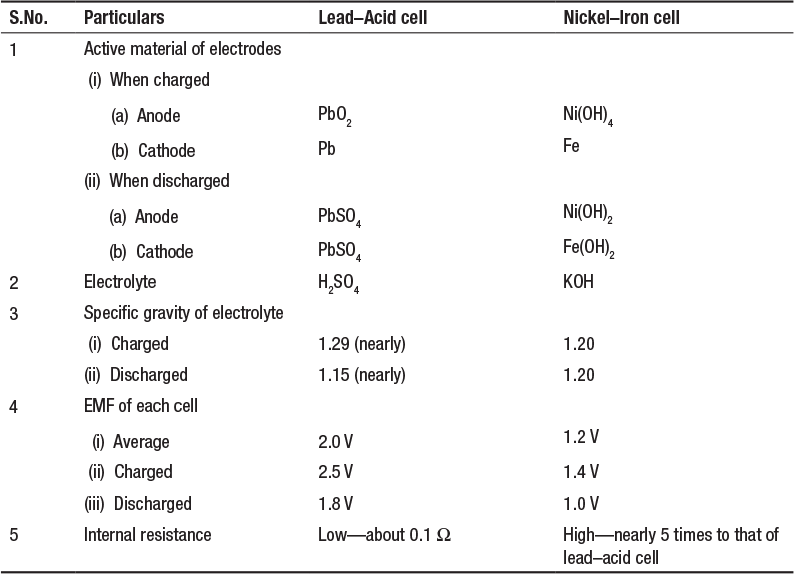
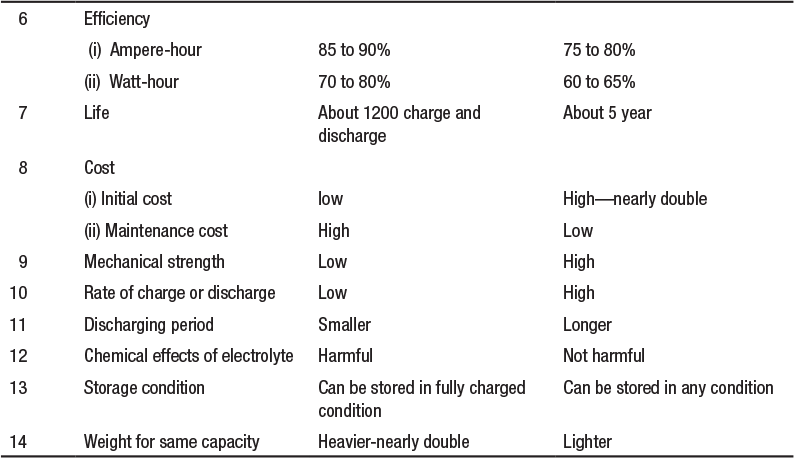
The two cells can be compared on the basis of the following particulars (Table 4.2):
Table 4.2 Comparison of Lead-Acid and Nickel-Iron Cell
4.15 NICKEL–CADMIUM CELL
It was developed by a Swedish scientist Waldemar Jungner in 1899.
4.15.1 Construction
| Cathode | – | Ni(OH)4 |
| Anode | – | Cd (Cadmium) |
| Electrolyte | – | KOH (Potassium hydroxide) of specific gravity 1.2. |
Its construction is similar to a nickel–iron cell with the difference that its extreme plates are positive instead of negative as in case of nickel–iron cell. Moreover, in this case, the positive plates are electrically connected to the container.
4.15.2 Chemical Action during Discharging
| At anode | – | Cd + 2OH → Cd(OH)2 |
| At cathode | – | Ni(OH)4 + 2K → 2KOH + Ni(OH)2 |
No change in the specific gravity of the electrolyte.
4.15.3 Chemical Action during Recharging
| At cathode | – | Ni(OH)2 + 2OH → Ni(OH)4 |
| At anode | – | Cd(OH)2 + 2K → Cd + 2KOH |
The two electrodes regain their original chemical composition.
4.15.4 Electrical Characteristics
- EMF—fully charged cell—1.4 V which decreases to 1.3 V rapidly. Average value 1.2 V which decreases to 1.0 V when fully discharged
- Internal resistance—very low, less than even lead–acid cell
- Efficiency—Ampere-hour: about 80%; watt-hour: about 65%
4.15.5 Advantages
- Very long active life nearly 20 years
- Can be stored in any condition, as there is no change in the specific gravity of the electrolyte.
- These cells can be charged in a short period (one hour)
4.15.6 Disadvantages
- It is very costly
- Low average emf and therefore, more cells are required for a particular voltage

Leave a Reply