The various advantages of nickel–cadmium cells such as longer life, low maintenance cost, low internal resistance, etc. prompted the scientists to develop these cells in small sizes. Since the emf developed by these cells is 1.2 V, which is very near to the emf developed by dry cells
(1.5 V), these cells have been developed by scientists of the same size as that of small carbon zinc or dry primary cells (called pencil cells). Today, these small-sized nickel–cadmium cells are used in cordless electric appliances, such as electric shavers, hearing aids, photography equipment, radios, tape recorders, and in space exploration.
The ingredients of a small nickel–cadmium cell are similar to that of a larger type. The plates are woven in the form of a screen, and a paste of active material is pressed into the spaces within the screen. A separator is placed between the positive and negative plate and then rolled in the form of a cylinder. The complete assembly is placed into a small can for protection. The negative plate is connected with the body of the can, which forms the negative terminal. An insulated metal button is placed at the top to which positive plate is connected and forms the positive terminal.
Although the initial cost of a nickel–cadmium cell is very high as compared to a carbon zinc primary cell (i.e., dry cell), but it is less expensive in the long run. This is because nickel–cadmium cell can be recharged and has very long life, whereas the dry cell (primary cell) cannot be recharged and has to be discarded when it is discharged.
4.16.1 Silver Button Cell
The constructional details of a silver oxide cell are shown in Figure 4.9. It contains cathode of silver oxide and anode of powdered zinc with an electrolyte of alkaline potassium hydroxide. These type of cells are generally made in button size (with typical dimensions of diameter 0.76 to 1.27 cm and thickness 0.2 to 1.5 cm). The working potential of this cell is 1.5 V.
Chemical reaction during discharging
AgO+ Zn + H2O → Ag + Zn(OH)2
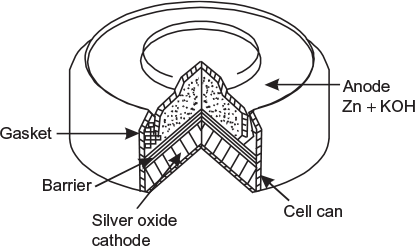
Fig. 4.9 Silver button cell
These cells are leak-proof (sealed), have very small internal resistance, deliver current at a constant voltage of 1.5 V, very handy, and occupy small space.
These cells are best suited for hearing aids, cameras, electronics watches, small electronic toys, and other electronic circuits.
4.17 SOLAR CELLS
A device that converts light energy (e.g., sun light) direct into electrical energy is called solar cell.
The construction of a simple solar cell is shown in Figure 4.10(a). A pure silicon (semiconductor) wafer is doped (the process of adding a suitable impurity to a pure semiconductor is called doping) with a specific amount of arsenic (donor impurity which has 5 electrons in its outermost orbit). This makes it an N-type semiconductor that contains excess of free electrons. The wafer is coated at its top with a very thin layer of silicon doped with appropriate amount of boron (acceptor impurity which has 3 electrons in its outermost orbit). This makes the top layer as a P-type semiconductor. Therefore, a contact surface becomes a P–N junction. A spot on the P-type layer and bottom of wafer (N-type material) are tinned for connecting the leads.
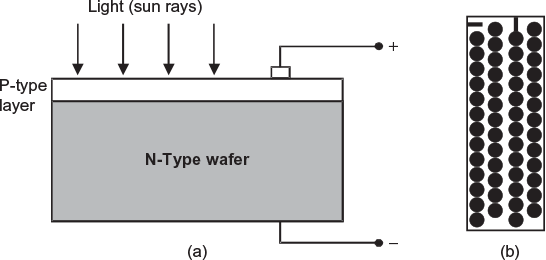
Fig. 4.10 Solar cell and Module (a) Solar cell (b) Solar module
When light (sun rays) falls on the top of P-type layer and penetrates into the N-type material just below it the free electrons in N-type material are activated and move across the P–N junction into P-type semiconductor. This continuous movement of charge carries (i.e., free electrons from N-type and holes from P-type) constitute electric current.
The operating voltage of one solar cell is about 0.39 V and the current varies between 30 to 40 mA. The power developed by a solar cell depends upon the exposed area and the intensity of light falling on its surface. Since it is difficult to have large silicon crystals heavy power cannot be developed. The maximum output power delivered by a solar cell, with sun light falling directly on a clear day is about 8 to 9 mW/cm2. To obtain higher voltages and current, a number of solar cells are connected in series parallel combination. A typical solar module having 52, 90 mm diameter silicon cells is shown in Figure 4.10(b). It is interesting to note that an 1100 × 42.5 cm module produces 31 W of power to charge a 12 V battery at 13.8 V and 2.25 A. The operating efficiency of a solar cell is very low (about 10%). Since they do not deteriorate when not in use, they have very long life (estimated to be thousands of years).
4.17.1 Applications
The major applications of solar cells are as follows:
- To charge nickel–cadmium batteries in satellites
- To provide power for calculators radio transistors, clocks, etc.
- To provide power to control devices, such as aperture control for movie cameras, microwave relay stations, etc.

Leave a Reply