You have no doubt been exposed to the concept of the ideal gas in chemistry and physics. Why go over ideal gases again? At least two reasons exist. First, the experimental and theoretical properties of ideal gases are far simpler than the corresponding properties of liquids and solids. Second, use of the ideal gas concept is of considerable industrial importance.
In this section we explain how the ideal gas law can be used to calculate the pressure, temperature, volume, or number of moles in a quantity of gas, and we define the partial pressure of a gas in a mixture of gases. We also discuss how to calculate the specific gravity and density of a gas. Then we apply the concepts to solving material balances.
7.1.1. Ideal Gas
The most famous and widely used equation that relates p, V, n, and T for a gas is the ideal gas law:

where p is the absolute pressure of the gas
V is the total volume occupied by the gas
n is the number of moles of the gas
R is the ideal (universal) gas constant in appropriate units
T is the absolute temperature of the gas
You can find values of R in various units inside the front cover of this book. Sometimes the ideal gas law is written as

Note that in Equation (7.1a) ![]() is the specific molar volume (volume per mole, V/n) of the gas. When gas volumes are involved in a problem,
is the specific molar volume (volume per mole, V/n) of the gas. When gas volumes are involved in a problem, ![]() will be the volume per mole and not the volume per mass. The inverse of
will be the volume per mole and not the volume per mass. The inverse of ![]() is the molar density, moles per volume. Figure 7.2 illustrates the surface generated by Equation (7.1a) in terms of the three properties p,
is the molar density, moles per volume. Figure 7.2 illustrates the surface generated by Equation (7.1a) in terms of the three properties p, ![]() , and T. Look at the projections of the surface in Figure 7.2 onto the two-parameter planes. The interpretation is as follows:
, and T. Look at the projections of the surface in Figure 7.2 onto the two-parameter planes. The interpretation is as follows:
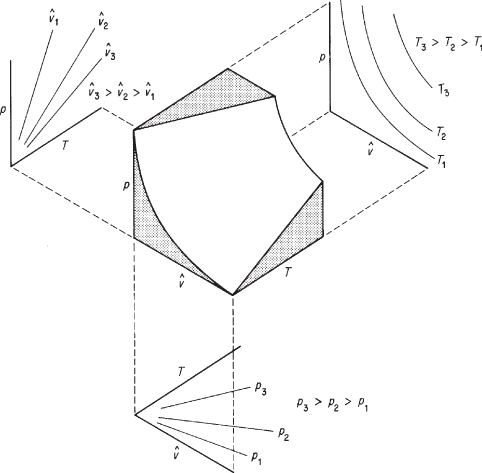
Figure 7.2. Representation of the ideal gas law in three dimensions as a surface
1. The projection to the upper left onto the p – T plane shows straight lines for constant values of ![]() . Why? Equation (7.1a) for constant specific volume reduces to p = (constant)(T), the equation of a straight line that passes through the origin.
. Why? Equation (7.1a) for constant specific volume reduces to p = (constant)(T), the equation of a straight line that passes through the origin.
2. The projection to the upper right onto the p – ![]() plane shows curves for values of constant T. What kinds of curves are they? For constant T, Equation (7.1a) becomes p
plane shows curves for values of constant T. What kinds of curves are they? For constant T, Equation (7.1a) becomes p![]() = constant, namely, a hyperbola.
= constant, namely, a hyperbola.
3. The projection downward onto the T – ![]() plane again shows straight lines. Why? Equation (7.1a) for constant p is
plane again shows straight lines. Why? Equation (7.1a) for constant p is ![]() = (constant)(T).
= (constant)(T).
For an ideal gas, Equation (7.1) can be applied to a pure component or to a mixture.
What are the conditions for a gas to behave as predicted by the ideal gas law? The major ones for a gas to be ideal are as follows:
1. The molecules do not occupy any space; they are infinitesimally small.
2. No attractive forces exist between the molecules so the molecules move completely independently of each other.
3. The gas molecules move in random, straight-line motion and the collisions between the molecules, and between the molecules and the walls of the container, are perfectly elastic.
Gases at low pressure and/or high temperature meet these conditions. Solids, liquids, and gases at high density—that is, high pressure and/or low temperature—do not. From a practical viewpoint, within reasonable error, you can treat air, oxygen, nitrogen, hydrogen, carbon dioxide, methane, and even water vapor, under most of the ordinary conditions you encounter, as ideal gases.
Several equivalent standard states known as standard conditions (S.C., or S.T.P., an acronym for “standard temperature and pressure”) of temperature and pressure have been specified for gases by custom. Refer to Table 7.1. Note that the standard conditions for the SI, Universal Scientific, and American Engineering systems are exactly the same conditions, but in different units. On the other hand, the natural gas industry uses a different reference temperature (15°F) but the same reference pressure (1 atm).
Table 7.1. Common Standard Conditions for the Ideal Gas
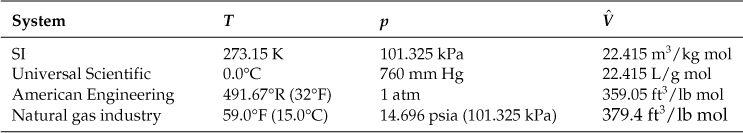
You can insert the values at S.C. into the ideal gas equation to calculate R in any units you want.
For example, what is R for 1 g mol of ideal gas with a volume in cubic centimeters, pressure in atmospheres, and temperature in kelvin?
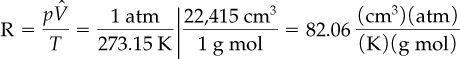
The fact that a substance cannot exist as a gas at 0°C and 1 atm is immaterial. Thus, as we shall see later, water vapor at 0°C cannot exist at a pressure greater than its vapor pressure of 0.61 kPa (0.18 in. Hg) without condensation occurring. However, you can calculate the imaginary volume at standard conditions, and it is just as useful a quantity in the calculation of volume-mole relationships as though it could exist. In what follows, the symbol V will stand for total volume and the symbol ![]() for volume per mole.
for volume per mole.
Because the SI, Universal Scientific, and AE standard conditions all refer to the same point in the p, ![]() , and T space, you can use the values in Table 7.1 with their units to change from one system of units to another. If you memorize the standard conditions, you will find it easy to work with mixtures of units from different systems.
, and T space, you can use the values in Table 7.1 with their units to change from one system of units to another. If you memorize the standard conditions, you will find it easy to work with mixtures of units from different systems.
The following example illustrates how you can use the standard conditions to convert mass or moles to volume. After reading it, see if you can explain to someone else how to convert volume to moles or mass.
Example 7.1. Use of Standard Conditions to Calculate Volume from Mass
Calculate the volume, in cubic meters, occupied by 40 kg of CO2 at standard conditions, assuming CO2 acts as an ideal gas.
Solution
Basis: 40 kg of CO2
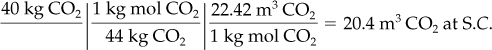
Notice in this problem that the information that 22.42 m3 of gas at S.C. = 1 kg mol of gas is applied to transform a known number of moles into an equivalent number of cubic meters. An alternate way to calculate the volume at standard conditions is to use Equation (7.1). Incidentally, whenever you report a volumetric value, you must establish the conditions of temperature and pressure at which the volumetric measure exists, since the term m3 or ft3, standing alone, is really not any particular quantity of material.
In many processes going from an initial state to a final state, you use the ratio of the ideal gas laws in the respective states and thus eliminate R as follows (the subscript 1 designates the initial state, and the subscript 2 designates the final state):
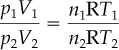
or
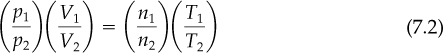
Note that Equation (7.2) involves ratios of the same variable. This result has the convenient feature that the pressures may be expressed in any system of units you choose, such as kilopascals, inches of Hg, millimeters of Hg, atmospheres, and so on, as long as the same units are used for both conditions of pressure (do not forget that the pressure must be absolute pressure in both cases). Similarly, the ratio of the absolute temperatures and ratio of the volumes result in dimensionless ratios. Note how the ideal gas constant R is eliminated in taking the ratios.
Let’s see how you can apply the ideal gas law to problems in the form of both Equation (7.2) and Equation (7.1).
Example 7.2. Application of the Ideal Gas Law to Calculate a Volume
Calculate the volume occupied by 88 lb of CO2 at 15°C and a pressure of 32.2 ft of water.
Solution
Examine Figure E7.2. To use Equation (7.2) the initial volume has to first be calculated as shown in Example 7.1.
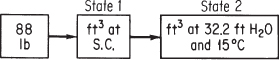
Figure E7.2
Then the final volume can be calculated via Equation (7.2) in which both R and (n1/n2) cancel. Table 7.1 does not list the pressure in feet of H2O at S.C. Where do you get the value? Look in Chapter 2 at the discussion of pressure, or calculate it (ρgh = p).
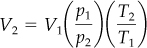
Assume that the given pressure is absolute pressure.
At S.C. (state 1) At p and T (state 2)
p = 33.91 ft H2O p = 32.2 ft H2O
T = 273 K T = 273 + 15 = 288 K
Basis: 88 lb of CO2
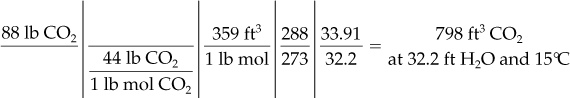
You can mentally check your calculations by saying to yourself: The temperature goes up from 0°C at S.C. to 15°C at the final state; hence, the volume must increase from S.C., and the temperature ratio must be greater than unity. Similarly, you can say: The pressure goes down from S.C. to the final state, so the volume must increase from S.C.; hence the pressure ratio must be greater than unity.
The same result can be obtained by using Equation (7.1). First obtain the value of R in the same units as the variables p, ![]() , and T. Look it up or calculate the value from p,
, and T. Look it up or calculate the value from p, ![]() , and T at S.C.:
, and T at S.C.:
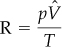
At S.C.: p = 33.91 ft H2O ![]() = 359 ft3/lb mol T = 273 K
= 359 ft3/lb mol T = 273 K
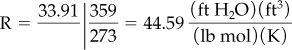
Now, using Equation (7.1), insert the given values, and perform the necessary calculations.
Basis: 88 lb of CO2
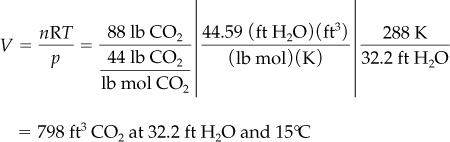
If you inspect the two solutions, you will observe that in both the same numbers appear, and that the results are identical.
To calculate the volumetric flow rate of a gas, ![]() , such as in cubic meters or cubic feet per second, through a pipe, you divide the volume of the gas passing through the pipe in a time interval by the value of the time interval. To get the velocity,
, such as in cubic meters or cubic feet per second, through a pipe, you divide the volume of the gas passing through the pipe in a time interval by the value of the time interval. To get the velocity, ![]() of the flow, you divide the volumetric flow rate by the area, A, of the pipe
of the flow, you divide the volumetric flow rate by the area, A, of the pipe
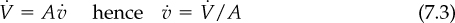
The (mass) density of a gas is defined as the mass per unit volume and can be expressed in various units, including kilograms per cubic meter, pounds per cubic foot, grams per liter, and so on. Inasmuch as the mass contained in a unit volume varies with the temperature and pressure, as we have previously mentioned, you should always be careful to specify these two conditions in calculating density. If not otherwise specified, the densities are presumed to be at S.C. As an example, what is the density of N2 at 27°C and 100 kPa in SI units?
Basis: 1 m3 of N2 at 27°C and 100 kPa
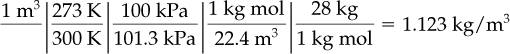
of N2 at 27°C (300 K) and 100 kPa
In addition to the mass density, sometimes the “density” of a gas refers to the molar density, namely, moles per unit volume. How can you tell the difference if the same symbol is used for the density?
The specific gravity of a gas is usually defined as the ratio of the density of the gas at a desired temperature and pressure to that of air (or any specified reference gas) at a certain temperature and pressure. The use of specific gravity occasionally may be confusing because of the sloppy manner in which the values of specific gravity are reported without citing T and p, such as “What is the specific gravity of methane?” The answer to the question is not clear; hence, assume S.C. for both the gas and the reference gas:
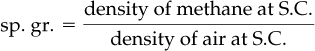

Leave a Reply