Frequently, as an engineer, you will want to make calculations for mixtures of gases instead of individual gases. You can use the ideal gas law, under the proper assumptions, of course, for a mixture of gases by interpreting p as the total absolute pressure of the mixture, V as the volume occupied by the mixture, n as the total number of moles of all components in the mixture, and T as the absolute temperature of the mixture. As the most obvious example, air is composed of N2, O2, Ar, CO2, Ne, He, and other trace gases, but you can treat air as a single compound in applying the ideal gas law.
Engineers use a fictitious but useful quantity called the partial pressure in many of their calculations involving gases. The partial pressure of Dalton, pi, namely, the pressure that would be exerted by a single component in a gaseous mixture if it existed alone in the same volume as that occupied by the mixture and at the same temperature as the mixture, is defined by

where pi is the partial pressure of component i in the mixture. If you divide Equation (7.4) by Equation (7.1), you find that
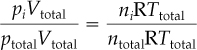
and
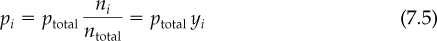
where yi is the mole fraction of component i. In air the percent oxygen is 20.95; hence, at the standard condition of 1 atm, the partial pressure of oxygen is pO2 = 0.2095(1) = 0.2095 atm. Can you show that Dalton’s law of the summation of partial pressures is true using Equation (7.5)?
Although you cannot easily measure the partial pressure of a gaseous component directly with commercial instruments, you can calculate the value from Equation (7.5) and/or Equation (7.6). To illustrate the significance of Equation (7.5) and the meaning of partial pressure, suppose that you carried out the following experiment with two nonreacting ideal gases. Examine Figure 7.2. Two tanks of 1.50 m3 volume, one containing gas A at 300 kPa and the other gas B at 400 kPa (both gases being at the same temperature of 20°C), are connected to an empty third tank (C) of the same volume. All of the gas in tanks A and B is forced into tank C isothermally. Now you have a 1.50 m3 tank of A + B at 700 kPa and 20°C for this mixture. According to Equation (7.5), you could say that gas A exerts a partial pressure of 300 kPa and gas B exerts a partial pressure of 400 kPa in tank C. Of course, you cannot put a pressure gauge on the tank and check this conclusion because the pressure gauge will read only the total pressure. These partial pressures are hypothetical pressures in tank C that the individual gases would exert if each was put into separate but identical volumes at the same temperature.
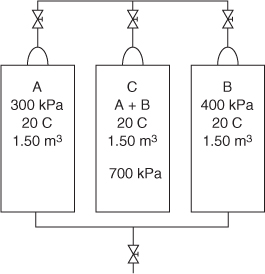
Figure 7.3. Illustration of the meaning of partial pressure of the components of an ideal gas mixture
When the $150 million Biosphere project in Arizona began in September 1991, it was billed as a sealed utopian planet in a bottle, where everything would be recycled. Its eight inhabitants lived for two years in the first large self-contained habitat for humans. But slowly the oxygen disappeared from the air—four women and four men in the 3.15 acres of glass domes eventually were breathing air with an oxygen content similar to that found at an altitude of about 13,400 ft. The “thin” air left the group members so fatigued and aching that they sometimes gasped for breath. Finally, the leaders of Biosphere 2 had to pump 21,000 lb of oxygen into the domes to raise the oxygen level from 14.5% to 19.0%. Subsequent investigation of the cause of the decrease in oxygen concluded that microorganisms in the soil that took up oxygen, a factor not accounted for in the design of the biosphere, were the cause of the problem.
Example 7.3. Calculation of the Partial Pressures of the Components in a Gas from a Gas Analysis
Few organisms are able to grow in solution using organic compounds that contain just one carbon atom such as methane or methanol. However, the bacterium Methylococcus capsulates can grow under aerobic conditions (in the presence of air) on C-1 carbon compounds. The resulting biomass is a good protein source that can be used directly as feed for domestic animals or fish.
In one process the off-gas analyzes 14.0% CO2, 6.0% O2, and 80.0% N2. It is at 300°F and 765.0 mm Hg pressure. Calculate the partial pressure of each component.
Solution
Use Equation (7.5): pi = ptotalyi.
Basis: 1.00 kg (or lb) mol of off-gas
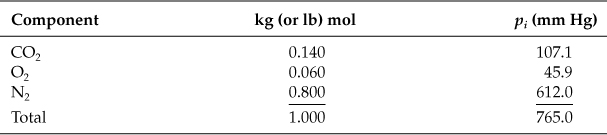
On the basis of 1.00 mol of off-gas, the mole fraction yi of each component, when multiplied by the total pressure, gives the partial pressure of that component. If you find that the temperature measurement of the flue gas was actually 337°F but the total pressure measurement was correct, would the partial pressures change? Hint: Is the temperature involved in Equation (7.5)?

Leave a Reply