Up to this chapter we have assumed that when a stream consists of several components, the total properties of the stream are the appropriately weighted sum of the properties of the individual components. For such ideal solutions, we could write down for the heat capacity of an ideal mixture, for example,Cpmixture=xACpA+xBCpB+xCCpC+…
or, for the enthalpy,ΔH^mixture =xAΔH^A+xBΔH^B+xCΔH^C+⋯
In particular, mixtures of gases have been treated as ideal solutions.
However, you must take into consideration other types of mixtures. You can prepare various kinds of binary solutions or mixtures:
- a. gas-gas
- b. gas-liquid
- c. gas-solid
- d. liquid-liquid
- e. liquid-solid
- f. solid-solid
You can ignore the energy changes that occur on mixing for cases a, c, and f. They are negligible. The other mixtures constitute real solutions.When a gaseous or solid solute(the compound to be dissolved) is mixed with a liquid solvent(the compound in which the solute is dissolved), the energy effect that occurs is referred to as the heat(really enthalpy) of solution.When a liquid is mixed with a liquid, the energy effect is called the heat (enthalpy) of mixing.The negative of the heat of solution or mixing is the heat (enthalpy) of dissolution.
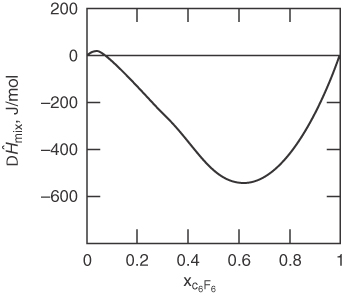
The heat of solution can be positive (endothermic) or negative (exothermic). Examine Figure 12.1, which shows the relative enthalpy values of a mixture of a fluorocarbon (C6F6) in benzene (C6H6).
You can treat heats of solution/mixing in the same way you treat chemical reactions. In the energy balance, you can (a) merge the heats of solution/mixing of the compounds in the system with the heats of formation, or (b) consolidate the effects of the heats of solution/mixing in one lumped term analogous to a heat of reaction term. For example, let us represent the solution of 1 g mol of HCl(g) into 5 g mol of by the following chemical equation:
HCl(g) +5H2O(l) →HCl[5H2O]
If you carry out experiments to measure the heat transfer from an apparatus at a constant 25°C and 1 atm by successively adding water to HCl, and arrange the experiments so that the energy balance reduces to Q = ΔH, then the values of ΔH would be the tabulation in the third column of Table 12.1. (The values incorporate a slight adjustment in the measured values of Q at the vapor pressure of the solution to adjust them to 1 atm, the standard state.) If you cumulate each incremental change in ΔH, you would obtain the fourth column in Table 12.1. Appendix I on the CD that accompanies this book contains other tables listing the heats of solution for common compounds. Table 12.1 shows that there are actually two concepts that incorporate the name “heat of solution”:
- a. The incremental (differential) heat of solution, column 3
- b. The integral heat of solution, column 4—the heat of solution for the combination of 1 mole of HCl(g) with n moles of H2O(I)
Usually “heat of solution” refers to concept b, and the enthalpy change is stated per mole of solute.
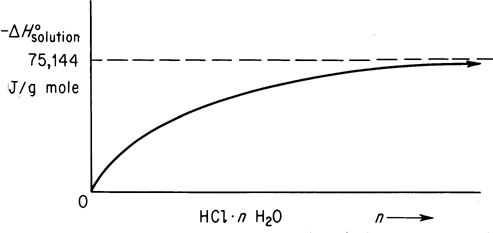
Figure 12.2 is a plot of the values listed in column 4 of Table 12.1. The asymptotic value of the heat of solution of HCl dissolved in an infinite amount of water is known as the heat of solution at infinite dilution (−75, 144 J/g mol HCl).
If you want to calculate the heat of formation of any of the solutions of HCl(g) in all you have to do is add the heat of solution to the heat of formation of HCl(g), as shown in column 5 in Table 12.1ΔH^f, solution o=ΔH^f,solute o+ΔH^solution o
(12.1)
where ΔH^solution o is the integral heat of solution at standard conditions per mole of HCl, and ΔH^f, solution o is the heat of formation of the solution itself per mole of HCl. It is important to remember that the heat of formation of the H2O does not enter into the calculation in Equation (12.1); it is defined as zero for the process of solution. Tables in reference books usually list data for the heats of formation of solutions in the standard state rather than the heats of solution themselves. In the following processes and examples, we assume that the systems are open, steady-state, or, if closed, that the accumulation term in the energy balance is ΔU = ΔH so that we discuss only enthalpies.
Table 12.1 Heat of Solution Data at 25°C and 1 atm
| Composition | Total Moles H2O Added to 1 mole HCl | ΔĤo for Each Incremental Step (J/g mol HCl) | Integral Heat of Solution (Cumulative ΔĤo) (J/g mol HCI) | Heat of Formation ΔH^fo (J/g mol HCI) |
|---|---|---|---|---|
| HCl(g) | 0 | −92,311 | ||
| HCl[1H2O(aq)] | 1 | −26,225 | −26,225 | −118,536 |
| HCl[2H2O(aq)] | 2 | −22,593 | −48,818 | −141,129 |
| HCl[3H2O(aq)] | 3 | −8033 | −56,851 | −149,161 |
| HCl[4H2O(aq)] | 4 | −4351 | −61,202 | −153,513 |
| HCl[5H2O(aq)] | 5 | −2845 | −64,047 | −156,358 |
| HCl[8H2O(aq)] | 8 | −4184 | −68,231 | −160,542 |
| HCl[10H2O(aq)] | 10 | −1255 | −69,486 | −161,797 |
| HCl[15H2O(aq)] | 15 | −1503 | −70,989 | −163,300 |
| HCl[25H2O(aq)] | 25 | −1276 | −72,265 | −164,576 |
| HCl[50H2O(aq)] | 50 | −1013 | −73,278 | −165,589 |
| HCl[100H2O(aq)] | 100 | −569 | −73,847 | −166,158 |
| HCl[200H2O(aq)] | 200 | −356 | −74,203 | −166,514 |
| HCl[500H2O(aq)] | 500 | −318 | −74,521 | −166,832 |
| HCl[1000H2O(aq)] | 1000 | −163 | −74,684 | −166,995 |
| HCl[50,000H2O(aq)] | 50,000 | −146 | −75,077 | −167,388 |
| HCl[∞H2O] | −67 | −75,144 | −167,455 |
SOURCE: National Bureau of Standards Circular 500, U.S. Government Printing Office, Washington, DC (1952).
You can treat a solution as a single compound in making calculations in an energy balance by using the property that enthalpies of solutions are state variables. One convenient procedure is to merge the heats of solution of a compound with the heats of formation, as indicated in column 5 of Table 12.1 and by Equation (12.1). Then the specific enthalpy of a solution relative to S.C. would beH^solution(T)=ΔH^f, solution o+[H^(T)−H^( S.C. )]solution
(12.2)
where the term in the brackets represents the sensible heat of the solution itself (any phase change is unlikely).
For example, suppose you want to find the ΔH^solution o that occurs at S.C. when a solution of 1 g mol of HCl dissolved in 1 g mol of H2O is placed in an infinite amount of water. Use the data listed in column 5 of Table 12.1, and subtract the enthalpy of the initial state from the enthalpy of the final state as follows:
(−167,455) −(−118,536) =−48,911 J/g mol HCl
As another example, consider a process in which a dilute solution of HCl is to be concentrated. Because enthalpy changes for heats of solution are state variables, you can easily look up the heats of formation of HCl solutions at their respective concentrations. Then you can calculate the enthalpy change between the final and initial states for a closed process, or the enthalpy change between the output and the input for a flow process. Thus, if you mix 1 mole of HCl[15H2O] and 1 mole of HCl[5H2O], you obtain 2 moles of HCl[10H2O], and the total enthalpy change at 25°C and 1 atm is
ΔHo=[2(−69,486)] −[1(−70,989) +1(−64,047)]
=−3936 J
You would have to remove 3936 J to keep the temperature of the final mixture at 25°C.
We should mention that the solution of a hydrated salt such as CaCl2· 6H2O requires a little care in the calculations. If you mix the hydrated salt with water or a CaCl2 solution, the procedure to calculate the enthalpy change is as follows: You first have to decompose (melt) the hydrated salt into a solid and water. Then you dissolve the total salt available into the total water available after the melting and solution. For example, from the data in problem 12.3, if 1 g mol of Na2CO3· 7H2O is dissolved in 8 g mol of the resulting solution contains 1 g mol of Na2CO3 and 15 g mol of H2O. The melting step involves the following enthalpy change:Na2CO3⋅7H2O(s) → Na2CO3(s) + 7H2O(l)ΔH^fo(kJ/gmol): −3201.18 −1130.92 −285.840
ΔHo=[7(−285.840) +1(−1130.92)] −[1(−3201.18)] = +69.38 kJ
The solution step isNa2CO3(s) + 15H2O(l) → Na2CO3[15H2O]ΔH^fo(kJ/gmol):−1130.92 0 −1163.70
ΔHo = (−1163.70) − (−1130.92) = − 32.78 kJ
The overall enthalpy change is 69.38 −32.78 =36.60 kJ.
Example 12.1 Application of Heat of Solution Data
You are asked to prepare an ammonium hydroxide solution at 77°F by dissolving gaseous NH3 in water. Calculate (a) the amount of cooling needed in British thermal units to prepare a 3.0% solution containing 1 lb mol of NH3, and (b) the amount of cooling needed in British thermal units to prepare 100 gal of a solution of 32.0% NH3.
Data: The following heat of solution data have been taken from NBS circular 500.
| Composition | State | −ΔH^fo (Btu/lb mol) | −ΔH^solno (Btu/lb mol) |
|---|---|---|---|
| g | 19,900 | 0 | |
| 1H2O | aq | 32,600 | 12,700 |
| 2H2O | aq | 33,600 | 13,700 |
| 3H2O | aq | 34,000 | 14,100 |
| 4H2O | aq | 34,200 | 14,300 |
| 5H2O | aq | 34,350 | 14,450 |
| 10H2O | aq | 34,600 | 14,700 |
| 20H2O | aq | 34,700 | 14,800 |
| 30H2O | aq | 34,700 | 14,800 |
| 40H2O | aq | 34,700 | 14,800 |
| 50H2O | aq | 34,750 | 14,850 |
| 100H2O | aq | 34,750 | 14,850 |
| 200H2O | aq | 34,800 | 14,900 |
| ∞H2O | aq | 34,800 | 14,900 |
Solution
The solution will be presented in abbreviated form.
Reference temperature: 77°F
- a. Basis: 1 lb mol NH3 ≡ 17 lb NH3wt % NH3 = lb NH3lb H2O + lb NH3(100)3=17(100)17+mH2O mH2O=550 lb or about 30 lb mol H2OFrom the table above, the ΔH^solno=−14,800 Btu/lb mol NH3, which is equal to 14,800 Btu removed from the system.
- b. Basis: 100 gal solutionFrom Lange’s Handbook of Chemistry, a 32.0% solution of NH3 has the following properties:Sp.gr.: NH30.889 H2O1.003The density of the 32.0% solution in pounds per 100 gal is(0.889)(62.4)(1.003)(100)7.48=744lb/100gal744(0.32)17=14.0 lb mol NH3/100 gal solutionBasis: 1 lb mol NH332.0=170017+mH2O mH2O=36 lb and nH2O=2 lb molCooling req’dBtu/100 gal}=(lbmolNH3100gal)(−ΔHsolnoBtulbmolNH3)Cooling req’dfor 100 gal32.0%NH3 soln}=(14.0)(−13,700)=−191,000Btu/100 galsoln(heat removed)
Self-Assessment Test
Questions
1. Define and show on a sketch (a) the integral heat of solution, and (b) the differential heat of solution.
2. Indicate whether the following statements are true or false:
- a. Heats of reaction and heats of solution represent the same physical phenomena.
- b. All mixtures have significant heats of solution.
- c. The heat of mixing at infinite dilution involves an infinite amount of solvent.
- d. Heats of solution can be positive and negative.
- e. A gas mixture is usually an ideal solution.
3. a. What is the reference state for H2O in the table for the heat of solution of HCl?
- b. What is the value of the enthalpy of the H2O in the reference state?
4. Repeat question 3 for HCl.
Problems
1. Calculate the heat of solution at standard conditions when 1 mol of a solution of 20 mol % HCl is mixed with 1 mol of a solution of 25 mol % HCl.
2. How much heat has to be added to a solution of 1 g mol of HCl in 10 g mol of H2O to concentrate the solution to 1 g mol of HCl in 4 g mol of H2O?
3. The heat of formation of H2SO4 is −811.319 kJ/g mol H2SO4. What is the heat of formation per gram mole of H2SO4 of a solution of 20% sulfuric acid?
Thought Problems
1. A tanker truck of hydrochloric acid was inadvertently unloaded into a large storage tank used for sulfuric acid. After about one-half of the 3000 gal load had been discharged, a violent explosion occurred, breaking the inlet and outlet lines and buckling the tank. What might be the cause of the explosion?
2. A concentrated solution (73%) of sodium hydroxide was stored in a vessel. Under normal operations, the solution was forced out by air pressure as needed. When application of air pressure did not work, apparently due to solidification of the caustic solution, water was poured through a manhole to dilute the caustic and free up the pressure line. An explosion took place and splashed caustic out of the manhole 15 ft into the air. What caused the incident?
Discussion Problem
A significant amount of energy is liberated when freshwater and saltwater are mixed. It has been calculated that the dilution of a cubic meter of freshwater per second in a large volume of seawater dissipates roughly 2.3 MW of power. If this energy could be put to use rather than heating the ocean, it is estimated that the potential of the flow of the Columbia River would yield 15,000 MW. The technology to collect such potential has been proposed, namely, to use a selective membrane that lets certain molecules through but holds others back. Instead of separating water from saltwater by imposing an electric potential on the membrane as in desalinization, the idea is to reverse the process and mix freshwater with saltwater to generate an electric current. The membranes are arranged so that positive ions flow in one direction and negative ions in the other direction. What do you think of the proposal?

Leave a Reply