Manufacturing standards, minimum slopes, permitted underlayment materials, and installation requirements for roofing materials are specified by building codes. The codes also regulate a roof’s required level of resistance to flame spread and fire penetration, tested according to standards ASTM E108 or UL 790 and rated as Class A, B, or C roof coverings (listed here in decreasing order of resistance). The International Building Code requires that roofs on buildings of Construction Types I, II-A, III-A, IV, or V-A meet at least Class B requirements and those on buildings of Construction Types II-B, III-B, and V-B meet at least Class C requirements (see Figure 1.3 and related text for an explanation of construction types). Roofs for single-family homes and other small residential or utility buildings generally may be nonclassified, except that where portions of such roofs are located close to property lines, a minimum Class C rating is required. Property insurance policies or local building regulations such as may apply in dense urban areas or in areas prone to wildfire may also impose roof class rating requirements.
Roof class rating tests apply to whole roof assemblies, including the membrane, shingles or other covering, underlayments, insulation, decking, and ballast, if any. Where a rated roof is required, the manufacturer of the roof covering should be consulted to determine the precise materials and construction requirements for meeting the classification. Broadly speaking, most low-slope roof membranes and noncombustible tiles (such as concrete or clay) can meet Class A requirements, as can some metal roof coverings and asphalt shingles made of glass felts; asphalt shingles made of organic felts and other metal roof coverings can meet Class B requirements; and fire-retardant wood shingles and shakes can meet Class C requirements.
FIGURE 16.58 A house is both roofed and sided with Red cedar shingles to feature its sculptural qualities. (Architect: William Isley. Photo by Paul Harper. Courtesy of Red Cedar Shingle and Handsplit Shake Bureau)

BUILDING ENCLOSURE ESSENTIALS: DISSIMILAR METALS AND THE GALVANIC SERIES
When different metals come into contact in the presence of moisture or some other conducting medium, electric current flows between them. Such metal pairs, or galvanic couples, form the basis, for example, for the storage of electrical energy in batteries. In building construction, galvanic couples are important for the manner in which the exchange of electrons affects the corrosion rates of the metals involved—a phenomenon that, if applied properly, can be used to positive effect or, if ignored, can lead to the premature deterioration of critical building components.
The Galvanic Series
In any galvanic couple, the metal donating electrons, called the anode, experiences an accelerated rate of corrosion; conversely, the metal receiving electrons, called the cathode, experiences corrosion at a reduced rate. This effect is relatively great for pairs of metals with large differences between their electrochemical potentials and proportionally smaller for those with lesser potential difference.
To facilitate the prediction of the corrosion potential of metal pairs, metals may be listed in a galvanic series, that is, in order of their relative electrochemical potential, with the most anodic, or active, metals at one end of the list and the most cathodic, noble or passive, ones at the other. Metal pairs positioned far apart in the series have greater corrosion potential than pairs closer to each other. Any number of galvanic series can be compiled for a group of metals in different conducting mediums and under different environmental conditions. For building construction, a galvanic series based on metals immersed in flowing sea water is used (Figure A).
Applying the Galvanic Series to Problems in Building Construction
Corrosion in the Anode and the Cathode
In Figure A, any metal listed higher in the series behaves as the anode when paired with any other metal listed lower in the series, which behaves as the cathode. Furthermore, in any such galvanic couple, the anode experiences accelerated corrosion, while the cathode’s rate of corrosion is reduced.
A common application of galvanic couples is in the use of anodic metals as protective coatings for other, more cathodic metals. For example, consider galvanized (zinc-coated) steel fasteners. Zinc appears higher in the galvanic series and is therefore the anode in this pair of metals. Even if the zinc coating is damaged and the steel itself becomes directly exposed to moisture and air, the steel can remain protected. As long as there is sufficient surrounding zinc to sustain galvanic action between the metals, only the zinc will corrode, in effect sacrificing itself to preserve the steel. For the same reason, zinc and zinc-aluminum alloys are frequently applied as protective metallic coatings to steel sheet used for roofing and flashing. These alloys form sacrificial, protective coatings for the underlying steel.
Environmental Conditions
Corrosion rates of metals are affected by the environment. Exterior metals exposed to marine, salt-laden air or to atmospheres laden with industrial pollutants will corrode at a faster rate than metals in less aggressive or more protected environments. The more severe the environment, the more important it is to avoid contact between dissimilar metals. Where dissimilar metals must be combined in such environments, metal pairs with the least electrochemical potential difference, that is, as close to each other as possible in the galvanic series, are preferred.
Relative Surface Area of Anode to Cathode
The corrosive effects within a galvanic couple are proportional to the relative surface areas of the metals involved. For example, consider copper roofing fastened with stainless steel fasteners. In this metal pair, copper is the anode and at risk of corrosion. However, the relative surface area of anode to cathode, that is, the surface area of the copper roofing, is very large in relation to the surface area of the stainless steel fasteners. This means that the electrochemical effects are diluted over a very large area of the copper and are therefore relatively weak. In fact, the use of stainless steel fasteners for the attachment of most types of metal roofing, including copper, is quite common. On the other hand, consider the use of galvanized steel fasteners for the attachment of the same copper roofing. In this case, the fasteners are the anode in the galvanic couple, and the ratio of surface areas of anode (fastener) to cathode (roof metal) is very low. This condition is likely to lead to greatly accelerated corrosion of the fasteners and is not recommended. As a general rule, when dissimilar metals are paired, the ratio of surface areas of anode to cathode should be kept as large as possible.
Cathodic Fasteners
As a corollary to the previous rule, where fasteners of one metal are used to attach metals of another type, the fasteners should be the cathodic metal in the pair. Since fasteners are typically small in surface area in comparison to the materials they attach, this approach ensures a relatively large anode-to-cathode surface area ratio. In practice, stainless steel fasteners are normally considered suitable for fastening construction metals of virtually all other types. This is a safe practice since stainless steel is the most cathodic metal normally found on the building site.
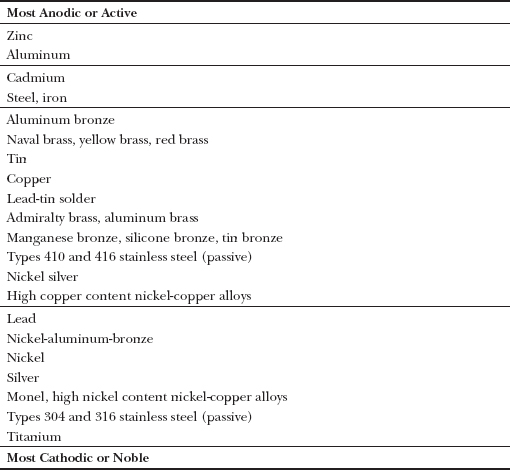
FIGURE A A galvanic series lists metals in order of electrochemical potential for a given conducting medium. The series shown here is for metals immersed in flowing sea water, adapted from ASTM G82. When paired as galvanic couples, metals positioned higher on the list will sacrificially corrode while protecting metals positioned lower on the list. The horizontal lines in the list separate metals into groups with relatively equal potential difference within each group. For example, although aluminum bronze and high copper content nickel-copper alloys are separated by eight other metals in this list, their electrochemical difference, and their relative corrosion potential as a galvanic couple, are roughly only as great as those between lead and titanium (separated by five other metals) or between cadmium and steel (which appear directly after each other).
Some metals, for example stainless steel, can appear in different locations within the series, depending on their electrochemical condition and the environment in which they are placed. In this series, stainless steels are listed in their so-called passive states, their expected state under conditions of normal architectural usage.
Water Flowing from One Metal to Another
Where rainwater flows from one metal type to another, the first metal should be anodic in relation to the second. Consider again a copper roof, in this case draining into a stainless steel gutter. As rainwater washes off the roof metal, it will carry dissolved copper ions into the gutter. Since the stainless steel is cathodic in relation to the copper, the gutter metal itself will remain unharmed. Conversely, consider the same roof with a galvanized steel gutter. In this case, the galvanized steel gutter metal is anodic in relation to the dissolved copper washing through it, and it may experience accelerated corrosion.
Insulating Dissimilar Metals
For galvanic action to occur, electric current must flow between the dissimilar metals. Thus, by insulating dissimilar metals from each other, galvanic corrosion can be prevented. For example, a layer of felt paper or rubberized asphalt membrane may be used to separate dissimilar sheet metal materials used in roofing and flashing work.
Paints, aluminum anodizing, and other coatings can also frequently act as insulators between one metal and another. However, when using coatings to separate dissimilar metals, the metals to be coated should be chosen carefully. Where an insulating coating is applied only to the anodic metal in a metal pair, that metal is at risk of accelerated corrosion if the coating becomes damaged. This is because if the coating fails in only limited areas, the exposed area of anodic metal is relatively small, and the galvanic action becomes concentrated in these limited areas (in other words, the anode-to-cathode surface area ratio is very small). For this reason, where coatings are used to separate dissimilar metals, it is safer to coat only the cathodic metal in the pair or to coat both metals.

FIGURE 16.59 A standing-seam metal roof with beautifully detailed overhangs, designed by architects Kallmann and McKinnell. (Photo by Steve Rosenthal)

SELECTED REFERENCES
1. National Roofing Contractors Association. The NRCA Roofing Manual: Membrane Roof Systems, The NRCA Steep-Slope Roofing Manual, The NRCA Architectural Sheet Metal Manual, The NRCA Spray Polyurethane Foam-Based Roofing Manual, and The NRCA Green Roof Systems Manual. Rosemont, IL.
These manuals, all updated regularly, are the most comprehensive guides to current U.S. practice for both low-slope and steep-slope roofing systems. The treatment is exhaustive, and both diagrams and text are excellent. These products are updated regularly and may also be available in combined editions.
2. Sheet Metal and Air Conditioning Contractors National Association. Architectural Sheet Metal. Chantilly, VA, updated regularly.
Architectural sheet metal roofs are copiously detailed in this excellent reference, along with every conceivable flashing, fascia, gravel stop, and gutter for flat and shingled roofs.
3. Zahner, L. William. Architectural Metals: A Guide to Selection, Specification, and Performance (3rd ed.). John Wiley & Sons, Hoboken, NJ, 1995.
This is one of the most comprehensive treatments of architectural metal types and their uses in construction available.
3. ASHRAE. ASHRAE Handbook – Fundamentals. American Society of Heating, Refrigerating, and Air-Conditioning Engineers, Atlanta, GA, updated regularly.
This handbook provides a definitive treatment of the physics of heat and moisture transfer through building assemblies and the application of these principals to building construction methods.

Leave a Reply