roadly, the refrigeration systems can be categorized as
- Air-refrigeration system.
- Vapour compression refrigeration system.
- Absorption refrigeration system.
8.4.1 Air-refrigeration System
Reversed Carnot cycle
Reversed Carnot cycle is shown in Figure 8.2. It consists of the following processes.

Figure 8.2 P−V and T−S Diagrams for Reversed Carnot Cycle
Process a–b: Absorption of heat by the working fluid from refrigerator at constant low temperature TL during isothermal expansion.
Process b–c: Isentropic compression of the working fluid with the aid of external work. The temperature of the fluid rises from TL to TH.
Process c–d: Isothermal compression of the working fluid during which heat is rejected at constant high temperature TH.
Process d–a: Isentropic expansion of the working fluid. The temperature of the working fluid falls from TH to TL.
COP of Refrigerator

Practical use of the reversed Carnot cycle is not possible for refrigeration purpose as the isentropic process requires very high speed operation, whereas the isothermal process requires very low speed operation.
Bell–Coleman Cycle or Reversed Brayton Cycle
In this system, air is taken into the compressor from atmosphere and compressed and then the hot compressed air is cooled in heat exchanger up to the atmospheric temperature. The cooled air is then expanded in an expander. The temperature of the air coming out from the expander is below the atmospheric temperature due to isentropic expansion. The low temperature air coming out from the expander enters into the evaporator and absorbs the heat. The cycle is repeated again and again. The working of reversed Brayton cycle is represented on P−V and T−S diagrams in Figure 8.3.

Figure 8.3 Reversed Brayton Cycle
Process 1−2: Suction of air into the compressor.
Process 2−3: Isentropic compression of air by the compressor.
Process 3−4: Discharge of high pressure air from the compressor into the heat exchanger. (The reduction in volume of air from V3 to V4 is due to the cooling of air in the heat exchanger).
Process 4−1: Isentropic expansion of air in the expander.
Process 1−2: Absorption of heat from the evaporator at constant pressure and suction of air into the compressor.

Work done per kg of air for the isentropic compression process 2−3 is given by,

Work developed per kg of air for the isentropic expansion process 4−1 is given by,

Net refrigerating effect per kg of air is given by,

For perfect inter-cooling, the required condition is T4 = T2

For isentropic compression process 2−3 and for expansion process 5−6, we have

Advantages and Disadvantages of Air-refrigeration System
Advantages
- Air is a cheaper refrigerant and available easily compared to other refrigerants.
- There is no danger of fire or toxic effects due to leakage.
- The total weight of the system per ton of refrigerating capacity is less.
Disadvantages
- The quantity of air required per ton refrigerating capacity is far greater than other systems.
- The COP is low and hence maintenance cost is high.
- The danger of frosting at the expander valves is more as the air taken into the system always contains moisture.
Example 8.1: Carnot refrigeration cycle absorbs heat at 250 K and rejects heat at 300 K.
- Calculate the coefficient of performance of this refrigeration cycle.
- If the cycle is absorbing 1,050 kJ/min at 250 K, how many kJ of work is required per second.
- If the Carnot heat pump operates between the same temperatures as the above refrigeration cycle, what is the coefficient of performance?
- How many kJ/min will the heat pump deliver at 300 K if it absorbs 1,050 kJ/min at 250 K.
Solution:
Given: T1 = 250 K; T2 = 300 K
- Coefficient of performance of Carnot refrigeration cycle

- Work required per secondLet WR = Work required per secondHeat absorbed at 250 K (i.e., T1), Q1 = 1,050 kJ/min= 17.5 kJ/s

- Coefficient of performance of Carnot heat pump

- Heat delivered by heat pump at 300 KLet Q2 = Heat delivered by heat pump at 300 K.Heat absorbed at 250 K (i.e., T1),Q1 = 1,050 kJ/min (given)

Example 8.2: The capacity of a refrigerator is 150 TR when working between − 5 and 20°C. Determine the mass of ice produced per day from water at 20°C. Also find the power required to drive the unit. Assume that the cycle operates on reversed Carnot cycle and latent heat of ice is 336 kJ/kg.
Solution:
Given: Q = 150 TR; T1 = −5°C = −5 + 273 = 268 K; T2 = 20°C = 20 + 273 = 293 K
Mass of ice produced per day
Heat extraction capacity of the refrigerator = 150 × 210 = 31,500 kJ/min
Heat removed from 1 kg of water at 20°C
to form ice at 0°C (ITR = 210 kJ/min) = Mass × Sq. heat × (Change in temperature) + hfg (ice)

8.4.2 Vapour Compression Refrigeration System
A simple vapour compression refrigeration system consists of the following equipments (Figure 8.4):

Figure 8.4 Schematic Diagram of Refrigeration Systems
- Compressor
- Condenser
- Expansion valve
- Evaporator
The low temperature and low pressure vapour is compressed by a compressor to high temperature and high pressure vapour. This vapour is then condensed into condenser at constant pressure and then passed through the expansion valve. Here, the vapour is throttled down to a low pressure liquid and passed through an evaporator, where it absorbs heat from the surroundings and vaporizes into low pressure vapour. The cycle then repeats again and again. The heat and work interaction in vapour compression process takes place in following way:
- Compressor requires work (W). The work is supplied to the system.
- During condensation, heat QH the equivalent of latent heat of condensation, etc. is removed from refrigerant.
- During evaporation, heat QL equivalent to latent heat of vaporization is absorbed by the refrigerant.
- There is no exchange of heat during throttling process through the expansion valve as this process occurs at constant enthalpy.
Figure 8.5 shows a simple vapour compression refrigeration cycle on T−S diagram for different compression processes. The cycle works between temperatures T1 and T2 representing the condenser and evaporator temperatures, respectively. The thermodynamic process of the cycle A−B−C−D is given as follows:

Figure 8.5 T−S Diagram of Vapour Compression Refrigeration Cycle
Process B–C: Isentropic compression of the vapour from state B to C.
Process C–D: Heat rejection in condenser is at constant pressure.
Process D–A: An irreversible adiabatic expansion of vapour through the expansion value. The pressure and temperature of the liquid are reduced. The process is accompanied by partial evaporation of some liquid. The process is shown by dotted line.
Process A–B: Heat absorption in evaporator at constant pressure. The final state depends on the quantity of heat absorbed and same may be wet, dry or superheated.
COP of Vapour Compression Cycle

Heat extracted from system (i.e., absorbed in absorber) = Heat transfer during the process, A−B = refrigerating effect.

Heat rejected from condenser

Factors Affecting the Performance of Vapour Compression Refrigeration System
- Sub-cooling: By passing the liquid refrigerant from condenser through a heat exchanger through which the cold vapour at suction from the evaporator is allowed to flow in the reversed direction. This process sub-cools the liquid but superheats the vapour. Thus, the COP not improved though refrigeration effect is increased. But, by making use of enough quantity of cooling water so that the liquid refrigerant is further cooled below the temperature of saturation. In some cases, a separate sub-cooler is also used for this purpose. In this case, COP is improved.
- Superheating of vapour: If the vapour at the compressor entry is in the superheated state which is produced due to higher heat absorption in the evaporator, then the refrigerating effect is increased. However, COP may increase, decrease or remain unchanged depending upon the range of pressure of the cycle.
- Change in suction pressure: The decrease in suction pressure decreases the refrigeration effect and at the same time increases the work of compression. But, both the effects tend to decrease the COP.
- Change in discharge pressure: The increase in discharge pressure results in lower COP. Hence, the discharge pressure should be kept as low as possible depending upon the temperature of the cooling medium available.
- Effect of volumetric efficiency of compressor: The factors like clearance volume, pressure drop through discharge, and suction values, leakage of vapour along the piston and superheating of cold vapour due to contact with hot cylinder walls affect the volume of the vapour actually pumped by the compressor.The volumetric efficiency of a compressor is defined as
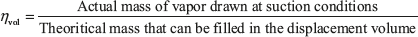
The actual amount of vapour sucked during the suction stroke is (V1 − V2) while the stroke volume is (V1 − VC). Volumetric efficiency decreases the refrigeration effect.
Comparison Between Vapour Compression Cycle and Reversed Carnot Cycle
- Vapour compression cycle requires more compression work than the Reversed Carnot cycle.
- No work is done during the throttling process in vapour compression cycle but work is done in expansion process in Reversed Carnot cycle.
- In vapour compression, expansion process is irreversible whereas in Reversed Carnot cycle, it is reversible.
Advantages and Disadvantages of Vapour Refrigeration Systems Over Air-refrigeration Systems
Advantages
- Vapour refrigeration system has higher C.O.P than the air-refrigeration system.
- Vapour refrigeration system has easer controllable expansion process.
- It has low running cost.
- It requires smaller evaporator.
Disadvantages
- Vapour refrigeration system has higher capital cost.
- In vapour refrigeration, system leakage problem may occur.
8.4.3 Absorption Refrigeration Cycle
The absorption cycle is a process by which refrigeration effect is produced through the use of two fluids and some quantity of heat input, rather than electrical input as in the more familiar vapour compression cycle. Both vapour compression and absorption refrigeration cycles accomplish the removal of heat through the evaporation of a refrigerant at a low pressure and the rejection of heat through the condensation of the refrigerant at a higher pressure. The method of creating the pressure difference and circulating the refrigerant is the primary difference between the two cycles. The vapour compression cycle employs a mechanical compressor to create the pressure differences necessary to circulate the refrigerant. In the absorption system, a secondary fluid or absorbent is used to circulate the refrigerant. Because the temperature requirements for the cycle fall into the low-to-moderate temperature range, and there is significant potential for electrical energy savings, absorption would seem to be a good prospect for geothermal application.
Absorption machines are commercially available today in two basic configurations. For applications above 32°F (primarily air conditioning), the cycle uses lithium bromide as the absorbent and water as the refrigerant. For applications below 32°F, an ammonia/water cycle is employed with ammonia as the refrigerant and water as the absorbent.

Leave a Reply