Specific heats of gases
Specific heat at constant pressure, cp. This is the quantity of heat energy supplied to raise 1 kg of the gas through 1°C or K, while the gas is at constant pressure.
Think of 1 kg of gas trapped in a cylinder. As heat energy is added, the pressure will rise. If the piston is allowed to move down the cylinder to prevent the rise in pressure, the amount of heat energy supplied to raise the temperature of the gas by 1°C is the specific heat of the gas at constant pressure.
Specific heat at constant volume, cv. This is the quantity of heat energy supplied to raise 1 kg of the gas through 1°C or K, while the gas is at constant volume.
Thinking again of the gas in the cylinder, in this case the heat energy is supplied while the piston is fixed, i.e. the volume is constant. The amount of heat energy added for the temperature to rise 1°C is the specific heat at constant volume, cv.
The specific heat of a gas at constant pressure is always a higher value than the specific heat at constant volume, because when the gas is receiving heat it must be allowed to expand to prevent a rise in pressure, and, while expanding, the gas is doing work in driving the piston down the cylinder. Extra heat energy must be supplied equivalent to the work done.
Note that this is an example of the equivalence of heat energy and work energy.
Example 2.1.3
1.5 kg of gas at 20°C is contained in a cylinder and heated to 75°C while the volume remains constant. Calculate the heat energy supplied if cv = 700 J/kgK.
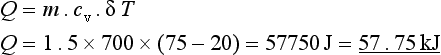
Example 2.1.4
A gas with a specific heat at constant pressure, cp = 900 J/kgK, is supplied with 80 kJ of heat energy. If the mass of the gas is 2 kg and its initial temperature is 10°C, find the final temperature of the gas if it is heated at constant pressure.
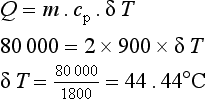

Leave a Reply