- The chemical reaction can be described by an overall stoichiometric relation as
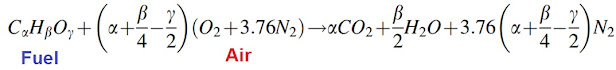
- The collection of elementary reactions that define the overall or global reaction is called a reaction or combustion mechanism.
- In intermediate stages, many chain initiation or chain branching reactions are involved
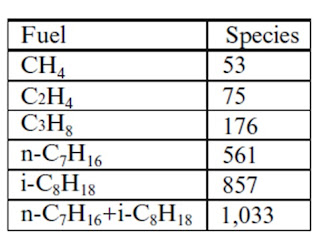
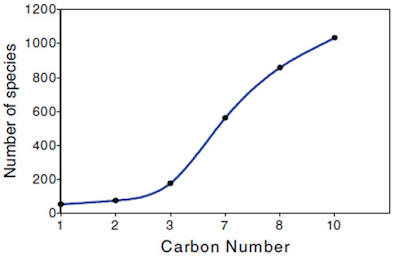
- The detailed combustion chemistry of hydrocarbon involves so many intermediate species and steps. Hence, only major species are considered for analysis or modeling

Leave a Reply