Properties of Mixtures
- The thermal properties of a pure substance are described by quantities including internal energy, u, enthalpy, h, specific heat, Cp, etc.
- Combustion systems consist of many different gases, so the thermodynamic properties of a mixture result from a combination of the properties of all of the individual gas species.
- The ideal gas law is assumed for gaseous mixtures, allowing the ideal gas relations to be applied to each gas component.
- Thermodynamic properties can be defined as either mass basis or mole basis

For more details click here:
Note: Refer to the book of “Thermodynamic” for mixture properties in reactive flows
Combustion Stoichiometry
- Air contains 21 mol percent O2 and 79 mol percent of N2 by volume
- For a given combustion device, say a piston engine, how much fuel and air should be injected in order to completely burn both?

- This question can be answered by balancing the combustion reaction equation for a particular fuel.
- A stoichiometric mixture has the exact amount of fuel and oxidizer such that after combustion is completed, all the fuel and oxidizer are consumed to form products.

- Combustion stoichiometry for general hydrocarbon fuel, CαHβOγ, with air, can be expressed

Methods of Quantifying Fuel and Air Content of Combustible Mixtures
- Fuel-Air Ratio (FAR): The fuel-air ratio, f, is given by

where mf and ma are the respective masses of the fuel and the air.
For a stoichiometric mixture
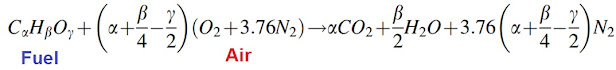
- The fuel-air-ratio (FAR) for above reaction is given as

where Mf and Mair (~28.84 kg/kmol) are the average masses per mole of fuel and air, respectively. The range of f is bounded by zero and one.
- Most hydrocarbon fuels have a stoichiometric fuel-air ratio, fs, in the range of 0.05–0.07
The air-fuel ratio (AFR);
- AFR is also used to describe a combustible mixture and is simply the reciprocal of FAR,
AFR = 1/FAR = 1/f
- For most hydrocarbon fuels, AFR is 14-20: that means 14–20 kg of air is required to achieve complete combustion of one kg fuel
Equivalence Ratio (ϕ):
- Normalizing the actual fuel-air ratio (FAR) by the stoichiometric fuel-air ratio provides the equivalence ratio,
ϕ = FARa/FARs

- The subscript, s denotes the values at the stoichiometric condition.
ϕ <1 is a lean mixture,
ϕ = 1 is a stoichiometric mixture,
ϕ >1 is a rich mixture.
Stoichiometric and Excess air
- The minimum amount of air that supplies the required amount of oxygen for the complete combustion of a fuel is called the stoichiometric or theoretical air
- The amount of air in excess of the stoichiometric air is called excess air. It is usually expressed in terms of the stoichiometric air as percent excess air. The amount of air less than stoichiometric is called a deficiency of air
- Percent Excess Air: The amount of air in excess of the stoichiometric amount is called excess air. The percent excess air, %EA, is defined as


Leave a Reply