Charles Coulomb, a French scientist, performed a number of experiments to see the effect of placing small charges near each other. From his experimental observations, he drew some conclusions and summed them up into two laws, known as Coulomb’s Laws of Electrostatics:
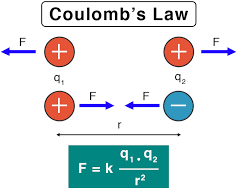
- First law relates the nature of force acting on the two charged bodies when placed near each other. It may be stated as like charges repel each other, whereas unlike charges attract each other.
- Second law tells us about the magnitude of the force exerted between two bodies when placed near to each other. It may be stated as the force exerted between two 1point charges:
- is directly proportional to the product of their strength;
- is inversely proportional to the square of the distance between them;
- depends upon the nature of medium in which the charges are placed.
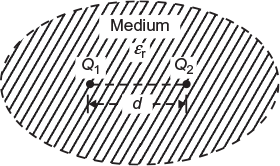
Fig. 3.1 Charges kept in a medium
Mathematically,
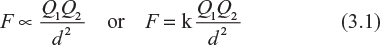
where
Q1 and Q2 = strength of two point charges or strength of the charge on two bodies in coulombs (see Fig. 3.1)
d = distance between the two charge points in metre, and
k = a constant whose value depends upon the medium in which the charges are placed and the system of units employed. In SI units, the value of k is given by
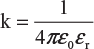
where ε0 = absolute permittivity of vacuum or air in SI units and its value is 8.854 × 10−12 F/m.
εr = relative permittivity of the medium in which the charges are placed. Its value depends upon the type of medium; for vacuum or air, its value is 1.
Now, the equation may be written as

The unit of force is Newton.
Here,
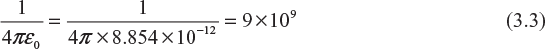
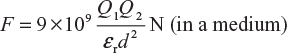
Therefore,

3.2.1 Unit Charge
In Equation (3.4), if we assume Q1 = Q2 = Q, d = 1 m, and F = 9 × 109 N, then
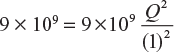
or
Q2 = 1
or
Q = ±1 C
Hence, 1 coulomb is that charge which when placed in air (strictly vacuum) at a distance of 1 m form an equal and similar charge that repels it with a force of 9 × 109 N.
Leave a Reply