The efficiency of a battery (or cell) can be defined in the following two ways:
- Quantity or A-H efficiency: The ratio of output ampere-hour during discharging to the input ampere-hour during charging of the battery is called quantity or ampere-efficiency of the battery.Mathematically,
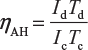 Where Id = discharging current in ampere;Td = discharging time in hour;Ic = charging current in ampere;Tc = charging time in hour.
Where Id = discharging current in ampere;Td = discharging time in hour;Ic = charging current in ampere;Tc = charging time in hour. - Energy or W-H efficiency: The ratio of output watt-hour during discharging to the input watt-hour during charging of the battery is called energy or watt-hour efficiency of the battery.Mathematically,
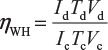 Where, Vd = Average terminal voltage during discharging,Vc = Average terminal voltage during charging
Where, Vd = Average terminal voltage during discharging,Vc = Average terminal voltage during charging
Example 4.4
An alkaline cell is discharged at a steady current of 4 A for 12 hour, the average terminal voltage being 1.2 V to restore it to its original state of voltage, a steady current of 3 A for 20 hour is required, the average terminal voltage being 1.44 V. Calculate the ampere-hour and watt-hour efficiencies in this particular case.
Solution:
Ampere-hour efficiency, 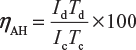
Where
Id = 4 A; Td = 12 hour; Ic = 3 A; Tc = 20 hour;
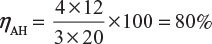
Where, Vd = 1.2 V; Vc = 1.44 V;
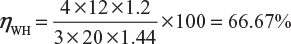
Example 4.5
A lead–acid cell has 17 plates, each 30 cm × 20 cm. The clearance between the neighbouring plates is 1 mm. If the resistivity of the acid is 1.6 ohm cm, find the internal resistance of the cell.
Solution:
Since there are 17 plates, the arrangement constitutes 16 tiny cells in parallel.
Resistance of each tiny cell, ![]()
Where ρ = 1.6 Ω cm; l = 0.1 cm; a = 30 × 20 = 600 cm2
∴
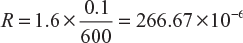
Total internal resistance of the cell, ![]()
Example 4.6
A discharged battery is put on charge at 5 A for 4 hour at a mean charging voltage of 13.5 V. It is then discharged in 6 hour at a constant terminal voltage of 12 V through a resistance of R ohm. Calculate the value of R for an ampere-hour efficiency of 85%.
Solution:
Ampere-hour efficiency, 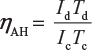
Where ηAH = 85%; Td = 6 hour; Ic = 5 A; Tc = 3.5 hour
∴
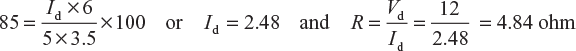
4.9 CHARGE INDICATIONS OF A LEAD–ACID BATTERY OR CELL
In batteries, the charge condition is determined by checking the specific gravity of the electrolyte (H2SO4). For a fully charged battery, the specific gravity of H2SO4 is 1.28 to 1.29. However, when the specific gravity falls below 1.15, the battery is fully discharged. In fact, to increase the life of battery, it should be recharged when the specific gravity of the electrolyte is found to be less than 1.18. The values of specific gravity for different condition of charge are given in Table 4.1.
Table 4.1 Specific Gravity V/s Charged Condition of a Lead-Acid Cell
| Specific Gravity | Condition |
|---|---|
| 1.280 to 1.290 | 100% charged |
| 1.230 to 1.250 | 75% charged |
| 1.190 to 1.200 | 50% charged |
| 1.150 to 1.160 | 25% charged |
| below 1.130 | fully discharged |
To check the specific gravity of H2SO4, an instrument called hydrometer is used (Fig. 4.7) which works on Archimedes principle. Commonly, the decimal point is omitted from the value of specific gravity, that is, 1.280 specific gravity, is spoken as 1280 and so on.
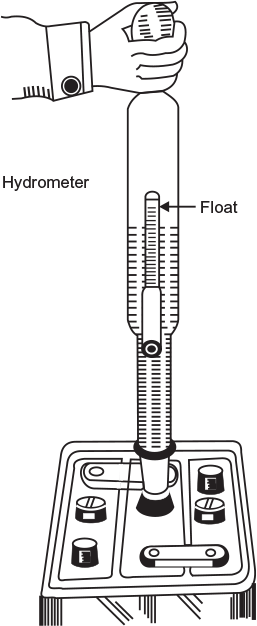
Fig. 4.7 Use of hydrometer to determine charged conditions of a lead-acid battery
However, the state of a battery can also be determined by checking the following:
- Voltage: When the terminal voltage of the battery on load is 2.1 to 2.5 V per cell, the battery is said to be fully charged. Whereas when the voltage of battery falls to below 1.8 V per cell, the battery is considered to be fully discharged and it is immediately put on charging.
- Colour of plates: When a lead–acid cell or battery is fully charged, its anode is PbO2 which is chocolate brown in colour and cathode is of Pb which is grey in colour. However, when the battery is fully discharged, both the plates attain PbSO4 as active material which is whitish in colour.

Leave a Reply