An electric device, such as battery, that converts chemical energy into electrical energy is known as electric cell.
4.2.1 Forming of a Cell
An electric cell essentially consists of the following:
- Two metal plates (electrodes) of different materials so that different potentials are built-up when chemical action takes place on them.
- A suitable solution (electrolyte) such as an acid, alkali, or salt solution—the solution must be capable to react chemically with the two electrodes.
When the two electrodes are immersed in a suitable electrolyte, different chemical actions take place on them and a potential difference is established between them.
4.2.2 EMF Developed in a Cell
The magnitude of emf of a cell depends upon the nature or material of the plates used as electrodes and the nature or type of electrolyte used.
4.3 TYPES OF CELLS
Broadly, electric cells may be divided into following two categories:
- Primary cells: The cells in which chemical action is not reversible are called primary cells, for example, voltaic cell, Daniell cell, Leclanche cell, dry cell, etc.In these cells, during discharging, one of the plates (usually negative plate) is consumed, which cannot be recovered by reversing the direction of flow of current through the cell. Therefore, chemical action in this case is not reversible and the cells cannot be recharged. This fact makes the primary cells rather an expensive source of electrical energy and rarely used in commercial applications.
- Secondary cells: The cells in which chemical action is reversible are called secondary cells, for example, lead–acid cell, nickel–iron alkaline cell (or Edison cell), nickel–cadmium–alkaline cell, etc.
In these cells, no electrode is consumed during discharging; however, the chemical composition of the plates is changed. When the direction of flow of current through the cell is reversed (charging), the plates and the electrolyte regain their original composition. Therefore, chemical action in this case is reversible, which is stored in the cell itself. For this reason, these cells are called storage cells or accumulators and are used in almost all commercial applications.
4.4 IMPORTANT TERMS RELATING TO AN ELECTRIC CELL
The following are the important terms related to an electric cell:
- Electromotive force (emf) of a cell: The energy supplied by a cell to one coulomb of charge is called emf of a cell. In general, it is defined as a potential difference (p.d.) between the two electrodes (positive and negative) of a cell on open circuit. It is represented by letter ‘E’.
- Internal resistance of a cell: The opposition offered to the flow of current by the internal composition of the cell itself is called its internal resistance. It is generally represented by letter ‘r’. Figure 4.1(a) shows a cell with its internal resistance.
- Terminal voltage: The potential difference across the terminals of a cell at load is known as its terminal voltage. It is generally represented by letter ‘V’.
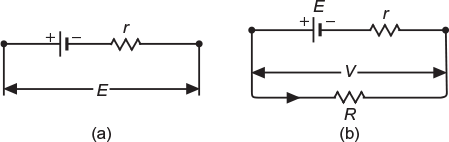
Fig. 4.1 Terminal voltage of a cell of no-lead and at load (a) Cell with internal resistance of no-load (b) Cell at load
When load is applied, it delivers current I to the external (load) resistor R as shown in Figure 4.1(b). The potential difference across the terminals reduces to V volts because of the voltage drop (I × r) in the internal resistance of the cell.
Therefore,

Leave a Reply