When a body, to which force is applied, moves in or opposite direction of the applied force, work is said to be done by or against the body.
Mathematically, Work = Force × distance or W = F ×d
Unit: The unit of work is Newton metre (Nm) or joule.
If, F = 1 N and d = 1 m; then, W = 1 Nm or joule.
Thus, when a force of 1 N applied on the body moves it to a distance of 1 m, the work done on the body is said to be 1 Nm or joule.
1.26 MECHANICAL POWER
The rate of doing work or the amount of work done per unit time is called power, that is,
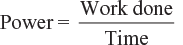
Unit: The unit of mechanical power is Newton metre per second (i.e., Nm/s) or joule/second (i.e., J/s).
However, the practical unit of mechanical power is horse power.
In fact, the rate of doing 75 kg m of work per second is known as one horse power.
1.27 HEAT ENERGY
The form of energy which produces a sensation of warmth is called heat.
Mathematically,
Heat,
H = m S θ
Where
m = mass of the body;
S = specific heat of the body; and
θ = rise or fall in temperature.
Unit: The unit of heat is kilocalorie (kcal)
If, m = 1 kg; θ = 1°C, and S = 1, that is, specific heat of water.
Then, H = 1 kcal
Hence, the amount of heat required to raise the temperature of 1 kg of water through 1°C is called one kilocalorie.
However, the smaller unit of heat energy is calorie.
One calorie is defined as the amount of heat required to raise the temperature of 1 gram of water through 1°C.
1 kilocalorie = 1000 calories
1.28 JOULES LAW OF ELECTRICAL HEATING
Joule (James Prescott Joule) established that there exists a definite relation between electrical energy expended and amount of heat produced. Thus, the relation is called Joule’s law of electrical heating.
This law stated that the amount of heat produced (H) is directly proportional to the electrical energy expended (W).
That is,
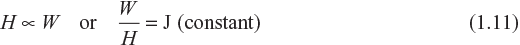
Where J is a constant called mechanical equivalent of heat and its value is determined as 4.18 joule per calorie (i.e., 1 calorie = 4.18 joule). It means that to produce one calorie of heat, 4.18 J of electrical energy is expended.
From equation (1.8), we get,
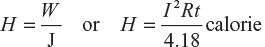
where I 2Rt is the electrical energy in joule.

Leave a Reply