When a lead–acid cell is ready for use, its positive plate is made of load peroxide (PbO2) chocolate brown in colour and the negative plate is of spongy lead (Pb) grey in colour. Both the plates are immersed in a dilute sulphuric acid (H2SO4) of specific gravity about 1.28. When a load is connected across the terminals of the cell, it starts delivering current to the load, and the process is called discharging of cell. In this process, the chemical energy stored in the cell is converted into electrical energy which is delivered to the load (external circuit).
Chemical action during discharging
The sulphuric acid when dissolved in water, its molecules are dissociated into hydrogen ions (2H+) and sulphate ions (SO4− −) which move freely (at random) in the electrolyte. When the load (resistor) is connected across the terminals as shows in Figure 4.6(a), the sulphate ions move towards the plate made of lead and hydrogen ions towards the plate made of lead peroxide. The following chemical action takes place in the two electrodes.
At lead plate
SO4− − − 2e …→ SO4 (radical)
The electrons travel through external load and after doing work reach at other electrode. The sulphate radical, chemically reacts with the electrode material (Pb) and forms lead sulphate (PbSO4).
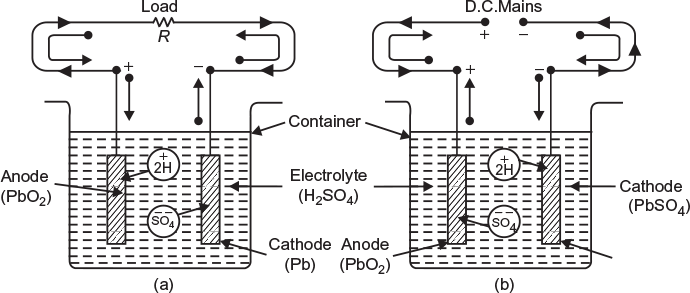
Fig. 4.6 Working of lead-acid cell (a) Working during discharging (b) Working during recharging
(Grey in colour) (whitish in colour)
At PbO2 plate
Each hydrogen ion (H+) on reaching the PbO2 plate takes one electron (the same electron which is given up by the sulphate ion at Pb state and has come to the other plate via external load resistor) from it to become hydrogen gas:
2H+ + 2e → 2H
(ion) (gas)
The hydrogen gas liberated at this electrode acts chemically with electrode material (PbO2) and reduces it to PbO which further reacts with H2SO4 forming PbSO4.
PbO2 + 2H → PbO + H2O
(Chocolate brown in colour)
PbO + H2SO4 → PbSO4 + H2O
(Whitish in colour)
Therefore, during discharging;
- Both the plates are converted to lead sulphate (PbSO4) that is whitish in colour.
- Sulphuric acid is consumed and water is formed which reduces the specific gravity of H2SO4 from 1.28 to 1.15.
- Terminal voltage per cell falls to 1.8 V form 2.0 V.
- Chemical energy is converted into electrical energy which is delivered to the load.
Chemical action during recharging
For recharging, position plate is connected to positive terminal of the source and negative plate is connected to the negative terminal as shown in Figure 4.6(b). During recharging, hydrogen ions (2H+) move towards the negative plate and sulphate ions (SO4− −) towards the positive plate. The following chemical actions take place at the two plates:
At positive plate
SO4− − − 2e → SO4 (radical)
SO4 + H2O → H2SO4 + O
The oxygen (in atomic state) reacts chemically with this plate material (PbSO4):
PbSO4 + O + H2O → PbO2 + H2SO4
(Whitish in colour) (Chocolate brown in colour)
At negative plate
Each hydrogen ion (H) on reaching the negative plate takes one electron (the same electron which is given up by the sulphate ion at positive plate and has come to negative plate via external circuit (i.e., source) from to become hydrogen gas;
2H+ + 2e → 2H
The hydrogen gas liberated at negative plate acts chemically with its material (PbSO4);
PbSO4 + 2H → Pb + H2SO4
(Whitish in colours) (Grey in colour)
Therefore, during recharging:
- Both the plates regain their original composition, that is, anode is converted back into lead peroxide (PbO2) and cathode into spongy lead (Pb).
- Water is consumed and sulphuric acid is formed which improves the specific gravity of H2SO4 from 1.15 to 1.28.
- Terminal voltage per cell increases from 1.8 V to 2.2/2.5 V.
- Electrical energy is converted into chemical energy which is stored in the cell.

Leave a Reply