Way back in 1922, when physicists were still studying the new and aston-
ishing properties of the basic constituents of matter, an experiment designed
15
16 Introduction to Quantum Physics and Information Processing
to measure the magnetic moment of atoms gave unexpected results. This was
the classic Stern–Gerlach experiment [36], designed to measure the magnetic
moments of atoms. The results brought out a new quantum property of an
electron, called intrinsic spin, which could take on quantized values, i.e., one
of two values only.
We can get a feel for the physics by looking at the classical definition of the
magnetic moment. The revolution of electrons around the nucleus of an atom
is like a circulating current, and a circulating current is a magnetic dipole.
The dipole moment ~µ equals the current times the area of the current loop,
with direction given by the axis about which the current circulates. When a
magnetic dipole is subjected to a non-uniform magnetic field
−→
B (~r), it feels a
force along the direction of change of the field:
−→
F =
−→
∇ (~µ ·
−→
B ), and will be
deflected. Measuring the deflection in a known magnetic field, the value of the
magnetic moment can be calculated.
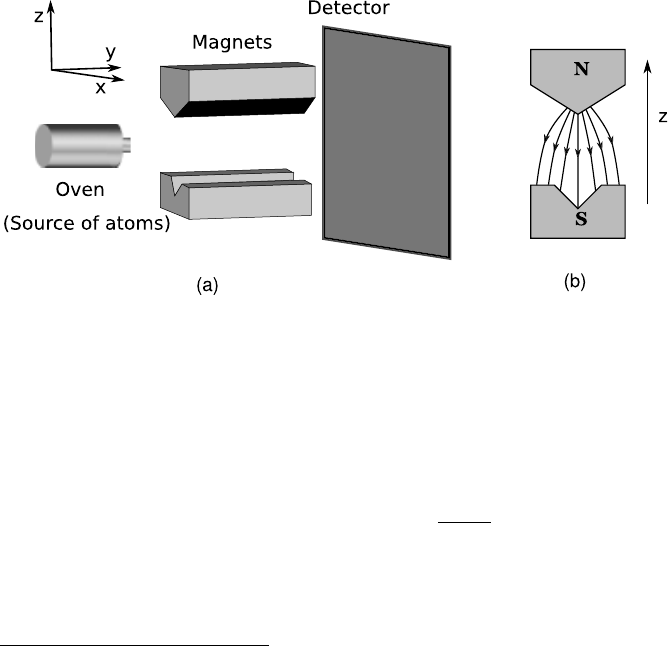
A schematic setup of the Stern–Gerlach type is shown in Figure 2.1.
N
N
SS
FIGURE 2.1: (a) The Stern–Gerlach Setup. (b) The inhomogeneous magnetic
field between asymmetric pole pieces.
The arrangement is such that in the region the electron beam passes
through, magnetic field is nearly constant in direction (taken to be ˆz)
1
but
has a strong z-dependent change in magnitude, i.e.,
−→
B ≈ B(z)ˆz. The force
on the dipole when placed in this field is
−→
F =
−→
∇ (~µ ·
−→
B ) =
−→
∇ (µ
z
B(z)) = µ
z
∂B(z)
∂z
ˆz. (2.1)
Thus the atom is deflected along the z-axis by an amount proportional to the
z-component of its magnetic moment. Remember: since a magnetic field can
deflect magnetic moments depending on their magnitudes and directions, it
1
By convention, in physics experiments, the coordinate system is aligned to the direction
of the magnetic field, which is always taken to be the z-axis.
A Simple Quantum System 17
can be used to select particular magnetic moments. The net magnetic moment
of a collection of atoms is just the vector sum of all the individual atomic
magnetic moments. A beam of atoms having a specific constant magnetic
moment along a particular direction is said to be polarized. It is possible to
produce a beam of polarized atoms by specific procedures.
The beam of silver atoms used in the original Stern–Gerlach experiment
was produced by heating silver in an oven. Each atom emerges with a random
direction of magnetic moment and the net magnetic moment is zero. If such
an unpolarized beam is sent into the non-uniform magnetic field, then since
each atomic magnetic moment is arbitrarily oriented, the z-component of the
magnetic moment could vary between ±µ. So we expect the beam to spread
between two extreme limits, which define the value µ of the magnetic moment
(see Figure 2.2(a)).
N
N
SS
N
N
SS
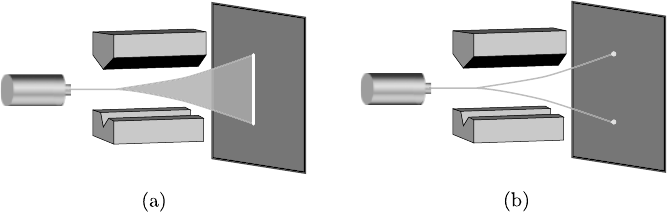
FIGURE 2.2: The Stern Gerlach experiment: (a) The classically expected
result. (b) What was actually observed.
Suppose, for simplicity, that this experiment is performed with a beam of
hydrogen atoms. The hydrogen atom consists of a single electron and a proton.
Classically one can think of the electron as orbiting the proton. The atom has
associated with it energy that has various contributions. At the first level, the
contribution depends on the electrostatic energy, determined by the distance
between the positive nucleus and the negative electron, i.e., the orbit radius.
Let’s assume that this is the minimum possible, or the “ground state” radius.
Second, the energy depends on the orbital velocity of the electron, contributing
to the orbital angular momentum of the atom. Finally, if the atom is subjected
to a magnetic field, its interaction with the field contributes to the energy. The
classical analogy however, is severely limited, because radius, velocity, and
component of magnetic moment along the magnetic field direction, all can
take continuous possible values whereas an atom’s energy is quantized: takes
on only certain discrete values. Correspondingly, the atom is said to exist in
possible quantized energy states.
The lowest energy state (s-state) has a symmetrical distribution of veloci-
ties such that there is no net circulating velocity. Therefore, in this state, the
atom is expected to have zero magnetic moment since the average “current”
is zero. This means that the beam, when it passes through the Stern–Gerlach
18 Introduction to Quantum Physics and Information Processing
setup, will just proceed without deflecting or spreading. The silver atoms used
in the original experiment also have zero average magnetic moment. The ex-
periment with hydrogen was also subsequently performed, in 1927 [56].
When the experiment was actually performed, there were two surprises.
The beam of atoms did not pass through undeflected. Nor did it spread, but
instead split into two beams symmetrically about the central axis, one up and
one down. Measuring the positions of the beams indicated a value of ±
1
2
,
in appropriate units,
2
for the magnetic moment! The appearance of Planck’s
constant h (numerically 6.63×10
−34
Js), in the magnitude, is a signature of the
quantum nature of this property. Whose magnetic moment? The atom in s-
state has no net magnetic moment, but has a lone electron. So this had to be an
intrinsic moment associated with the electron in the atom. Thus, the magnetic
moment of the electron is allowed only to take one of two discrete values!
Classically, the magnetic moment is proportional to the angular momentum of
the system. Here, the electron magnetic moment is proportional to a property
called intrinsic spin, which mathematically behaves like angular momentum.
Thus was discovered the spin of the electron, a quantum property that is
allowed only two possible values, plus or minus a half.
A word of caution is in place here. In the previous paragraphs we gave a
description of the atom in a classical way, to help you form a picture of the
physics involved. However, this description is severely limited. In truth, the
orbiting of the electrons about the nucleus is not like point particles revolving
in space. Nor is the electron really spinning about its axis, it is a point particle
with no extension in space! We want to emphasize that the electron spin is a
purely quantum mechanical concept, and is physically probed by virtue of its
interaction with a non-uniform magnetic field.
You can well imagine that the choice of a particular direction for the
magnetic field inhomogeneity cannot affect the value of the magnetic moment
of the electron: so even if the apparatus was tilted along any direction, the
results would remain the same.
Thus an atom with a single electron, described on the basis of its spin
alone, is a 2-state quantum system, well suited as a candidate qubit. We will
now illustrate the properties of a qubit using this experiment

Leave a Reply