We are studying classical gates to help us develop quantum gates. Quantum
gates are unitary. This means they are reversible: they can be “run backward”.
More practically, the meaning is that the inputs can be deduced from the
outputs. Most classical gates however, are irreversible, and cannot as such
be extended to quantum gates. For example, the AND gate, being 2 → 1 is
irreversible: it gives an output of 0 for more than one input set: (0, 0), (0, 1),
and (1, 0). So given only the output, the input cannot be deduced. So is the
OR gate and all the other famous 2-bit 2 → 1 gates! For an n-bit gate to be
reversible it must at least be a 1 → 1 mapping. Further it must give distinct
outputs for different inputs. Thus the outputs are all simply permutations
of the inputs. In terms of matrix representations, reversible gates must be
invertible. The classical two-bit gates represented by non-square matrices can
clearly not be inverted.
The idea of reversibility in classical computation has been studied long
before quantum gates were thought of (see for example Bennett [7]). It began
with the ideas of Landauer [46], who argued that erasure of information is
110 Introduction to Quantum Physics and Information Processing
accompanied by a loss in energy. Irreversible gates essentially erase some bits
of information in their functioning, and this should lead to intrinsic dissipation
of energy. Thus if one wants the most energy-efficient computing machine it
should employ reversible gates.
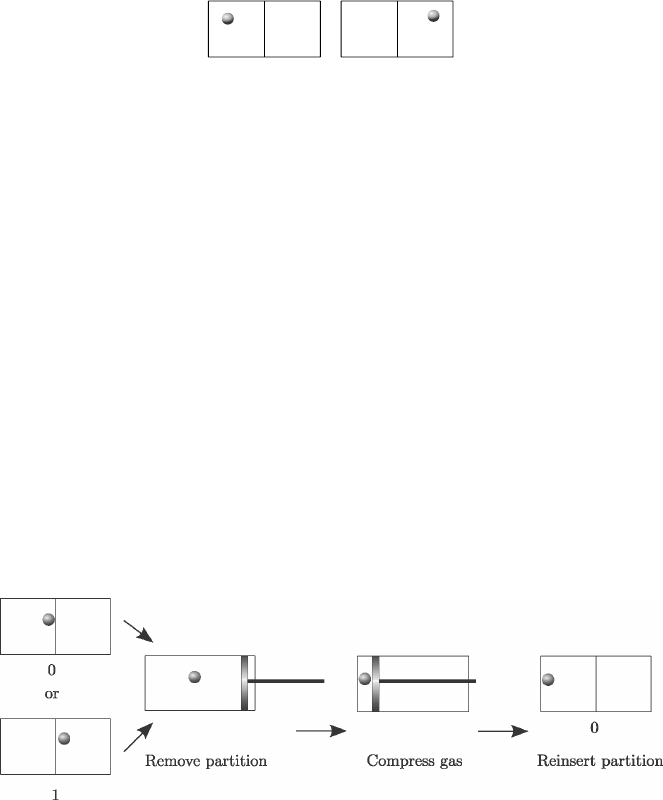
bit 0 bit 1
FIGURE 6.3: A simple thermodynamic system encoding a bit of information.
A simple way to understand how erasing information costs is in terms
of the thermodynamic quantity known as entropy. We will see more of this
concept when we study quantifying information. At present we want to see how
Landauer argued that information erasure causes an increase in the entropy
of the environment and therefore a decrease in the energy of the system. His
main point was that information was not something abstract, but was in fact
the physical system used to represent it. In an illustrative example due to
Szilard [68], a bit of information can be encoded in terms of the location of a
molecule in the left or right of a partition in a transparent box (Figure 6.3). If
we look at the box and find the molecule in the left partition then the system
encodes a logical 0, and if it is on the right side then it encodes a logical 1.
We can write one bit of information in this system by putting the molecule in
the appropriate half.
One way to erase the information contained in the location of the molecule
is to remove the partition and push the molecule to one end by compressing
the “gas” with a piston. If we then replace the partition, the system reads 0
irrespective of what was encoded in it initially (Figure 6.4).
FIGURE 6.4: Erasing a bit of information.
Thermodynamics tells us how to calculate the work done in this process.
The entropy of a thermodynamic system is related to the logarithm of the
number of microscopic states available to the system. Since the molecule could
be in one of two locations, the entropy associated with the single bit encoded
Leave a Reply