The essential components of a chemical engineering curriculum are presented in the sections that follow. The courses corresponding to each one of the four responsibilities are described first, followed by the description of engineering science and specialized fundamental chemical engineering courses and the basic science and mathematics courses that serve as the foundation for engineering education. The role and contribution of general education courses are also outlined.
3.3.1 Advanced Chemical Engineering Courses
The advanced chemical engineering courses are typically taught in the third and fourth (junior and senior) years of the program. Students are exposed to the concepts underlying reactor design, separations, transport processes, and process control. In addition to these courses, the students generally take several technical elective courses based on their interests. A final capstone design course that requires the students to synthesize the concepts from all courses is an essential requirement for all chemical engineering programs.
3.3.1.1 Chemical Reaction Engineering
The course listed most often as Chemical Reaction Engineering or Chemical Engineering Kinetics explains and teaches the design of chemical reactors. The essence of the design problem is explained simply as follows: It is desired to produce a certain quantity of a chemical determined by the market demand for that chemical. The engineer is required to specify the size of the vessel and the operating conditions for producing the chemical at the desired rate.
To solve this design problem, the engineer needs to understand the factors governing the rate of the reaction, which is generally defined in terms of the quantity reacting per unit time per unit measure of reactor size. The intrinsic kinetics of the reaction is dependent on the concentrations of species involved in the reaction as well as the temperature. Additional factors that play a role in determining the actual or observed rate in the reactor may include flow rate and pattern, agitation speed, and so on.
The course on reactor and reaction engineering teaches students the fundamental concepts and techniques related to reactor design. The desired outcome of the course is competency on the part of the student in design and analysis of chemical reactors for the different types of reactors employed in chemical processes. Following are some of the core concepts taught in the course:
• Determination of intrinsic kinetics: The student learns about quantifying the rate of reaction and factors determining this rate. The parameters in the rate expression (equation representing the relationship between the rate and factors affecting the rate) need to be determined experimentally, and the student learns about conducting laboratory experiments for this purpose, as well as analysis and interpretation of the data obtained.
• Design of reactors: The types of reactors used in the chemical industry can be categorized on the basis of a number of different criteria: whether they are operated in a batch mode, where conditions change with time, or in a continuous mode, where conditions are invariant and the system is at a steady state; whether the reactors operate under isothermal (constant temperature) or nonisothermal (temperature varying with time or position within the reactor) conditions; whether the reaction is homogeneous (single phase) or heterogeneous (two or more phases); and so on. Figure 3.3 shows the schematic sketches of batch and continuous reactors used in the chemical industry [7]. The operational characteristics of batch and continuous reactors are qualitatively and quantitatively different from each other. Design of batch reactors involves determining the batch size and time, whereas the design of continuous reactors involves determining the reactor size or residence time. Continuous reactors are further classified according to the flow pattern. Design equations for mixed flow reactors (contents well mixed) are different from segregated flow reactors (contents not intermixed). A large number of industrial reactions are fluid-solid heterogeneous reactions, the solid being a catalyst. Many other reactions involving a solid phase are noncatalytic; that is, the solid participates in the reaction. Each of these cases requires a distinct approach for reactor design, which a chemical engineering student must understand and master.
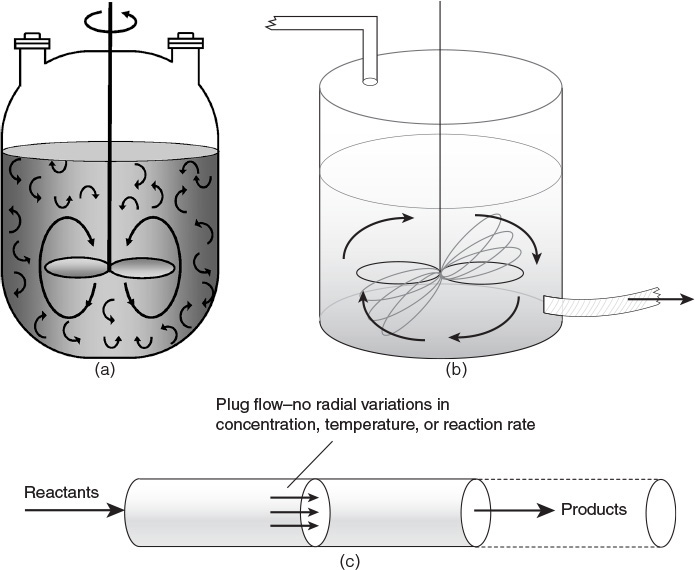
Figure 3.3 Batch and continuous reactors—(a) batch reactor, (b) continuous stirred tank reactor, (c) tubular plug flow reactor.
Source: Fogler, F. S., Elements of Chemical Reaction Engineering, Fourth Edition, Prentice Hall, Upper Saddle River, New Jersey, 2004.
• Analysis of flow characteristics: The mixed flow reactor and the segregated flow reactor represent ideal flow reactors. The actual flow pattern in operating reactors frequently does not conform to the idealized flow pattern. A student must learn to model the flow pattern and develop analytical techniques to identify the departure from ideality for accurately predicting the extent of reaction and implementing corrective measures.
The generalized concepts learned in this course enable a chemical engineer to apply the knowledge for any reactor system ranging from a single homogeneous reaction to multiple reactions in complex systems, such as enzyme-mediated reactions of complex substances in biological systems.
3.3.1.2 Separation Processes
Variously called Unit Operations or Mass Transfer Operations, the Separation Processes course deals with physical separations of components and is as critical to a chemical engineering student as the course on reactor design. This course teaches the students how to solve a variety of separation challenges:
• Separation of components from raw materials to obtain a reactor feed of desired purity
• Removal of undesirable contaminants from the raw product stream so that the required product specifications are met
• Recovery of valuable components from the waste stream before it is released to the environment
A student, after this course (or courses) on separations, will be able to make a decision regarding the type of separation process and design the equipment to accomplish the desired separation. Following are the most common separation techniques employed in the chemical industry:
• Distillation: Distillation is the most common technique utilized by the chemical industry for separating miscible components that exist in liquid phase at the given conditions. Distillation is based on differing volatilities (ease of vaporization) of the components of the mixture. For example, a distillation column used in petroleum refining allows us to separate gasoline from diesel (and aviation fuel and other components). Similarly, ethanol-water separation can be effected by distillation. Figure 3.4 shows a general schematic of a distillation column [8].
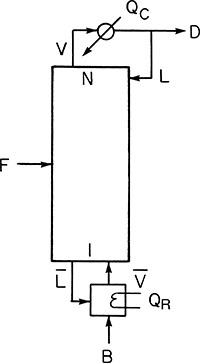
Figure 3.4 Schematic of a distillation column.
Source: Wankat, P. C., Separation Process Engineering, Third Edition, Prentice Hall, Upper Saddle River, New Jersey, 2012.
Feed F to the column is separated into distillate D and bottoms B. The energy needed for the separation is supplied in the reboiler at the bottom of the column to generate a vapor stream. This vapor stream is contacted with a liquid stream flowing down the column. A condenser located at the top of the column condenses the vapor at the top, returning part of the condensed liquid back to the column. The distillate product at the top is preferentially enriched in the more volatile components of the feed, whereas the bottom product is enriched in the less volatile components. Figure 3.5 shows a system of three distillation columns used for solvent recovery in a chemical plant.

Figure 3.5 A chemical plant with three distillation columns.
Source: Wankat, P. C., Separation Process Engineering, Third Edition, Prentice Hall, Upper Saddle River, New Jersey, 2012.
• Gas absorption/stripping: Both gas absorption and stripping operations involve transferring a component from one phase to another. Removing a contaminant gas (such as H2S or sulfur oxides) from a gas stream (prior to venting the stream to the atmosphere) by absorbing it in an alkaline solution is an example of gas absorption. Stripping is the reverse of absorption wherein a dissolved gas is stripped from a liquid. These operations are based on manipulating the affinity of the target component for one phase over the other. Figure 3.6 shows a schematic of an absorber-stripper system.
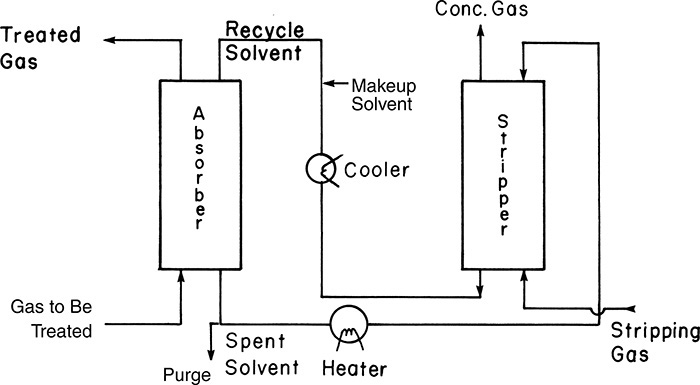
Figure 3.6 Absorber-stripper system.
Source: Wankat, P. C., Separation Process Engineering, Third Edition, Prentice Hall, Upper Saddle River, New Jersey, 2012.
The gas to be treated for the removal of a component is fed at the bottom of the absorber, where it is contacted with a solvent that preferentially dissolves the component. Solvent needed for absorption is regenerated in the stripping column, where the solvent exiting the absorption column that is enriched in the component is contacted with the stripping gas. The stripping column operates at a higher temperature; as a result, the component dissolved in the solvent is transferred to the gas phase.
• Liquid-liquid extraction: A liquid stream is often purified by transferring the impurity to another liquid stream that is immiscible with the feed stream. As with absorption/stripping, manipulating operating conditions allows an engineer to change the affinity of the component of interest. Typically, but not always, one liquid stream is aqueous while the other one is organic. Figure 3.7 shows a conceptual schematic of separation using liquid-liquid extraction using two different contacting devices.
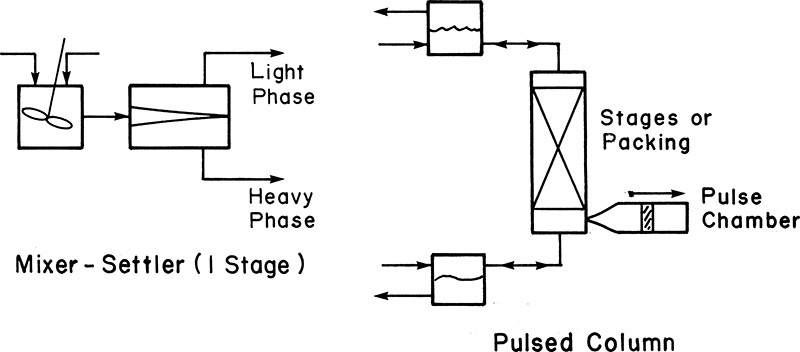
Figure 3.7 Liquid-liquid extraction.
Source: Wankat, P. C., Separation Process Engineering, Third Edition, Prentice Hall, Upper Saddle River, New Jersey, 2012.
In the mixer-settler unit shown on the left, the two immiscible phases are stirred together to promote mixing and transfer of the component of interest. The two phases are separated in the settler unit under the influence of gravity, taking advantage of the density differences. The light and dense phases are removed from different locations in the settler unit. The mixing of phases and the resultant transfer of the component is accomplished by providing a contact surface by means of a packing or stages in a pulsed column. Pulsing the column results in regeneration of surface, promoting the transfer of the component. The phases are separated, as shown in the figure.
• Adsorption: A component is removed from the fluid (gas or liquid) phase by contacting the fluid with a solid. The component is preferentially sorbed to the surface of the solid and essentially removed from the fluid. The principle of the pressure-swing adsorption cycle used for clean-up of an impurity containing gas is shown in Figure 3.8.
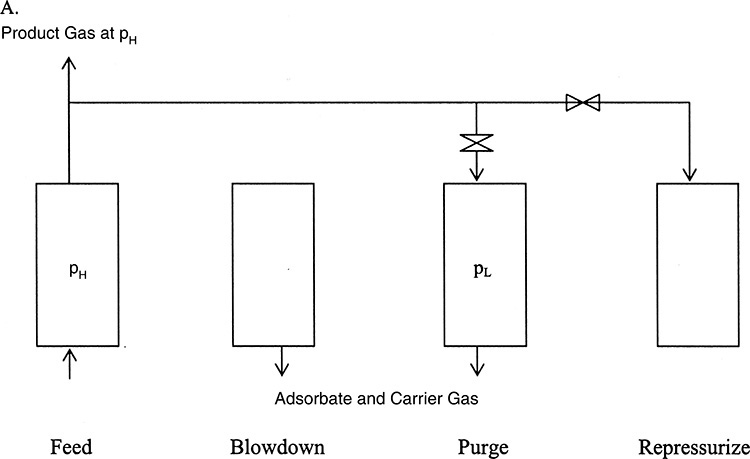
Figure 3.8 Principle of pressure-swing adsorption.
Source: Wankat, P. C., Separation Process Engineering, Third Edition, Prentice Hall, Upper Saddle River, New Jersey, 2012.
The gas containing the impurity is contacted with the adsorbent in a column at high pressure. The adsorbent preferentially sorbs the impurity removing it from the gas, which is collected at the product gas at high pressures (PH). The adsorption column operates in a batch mode; that is, the adsorbent gets progressively saturated with the impurity, losing the ability to clean the gas. At this point, the feed gas flow is stopped, and the column pressure lowered. The third stage consists of purging the column at this low pressure (PL), where the impurity picked by the adsorbent is desorbed or removed, regenerating the column capacity. The last stage is repressurizing the column to high pressure in order to repeat the cycle.
• Evaporation and drying: A stream dilute with respect to a solute is often subjected to evaporation for concentration. For example, a dilute caustic solution is the first product in the caustic soda (NaOH) manufacture. Progressively concentrated solutions (50%, 70%, etc.) are obtained through evaporation. Drying involves continuing this solvent removal to its extreme limit, leaving behind only the pure solute. The solvent can be aqueous (as in the majority of these operations) or organic. These operations consume significant amounts of energy, and an improperly designed evaporator or dryer can impact the plant economics adversely.
• Membrane separations: Figure 3.9 illustrates the principle of separations using membranes. Membranes are semipermeable barriers between two phases, and the separation of a mixture is based on the preferential transfer of one or more of the components across this barrier.
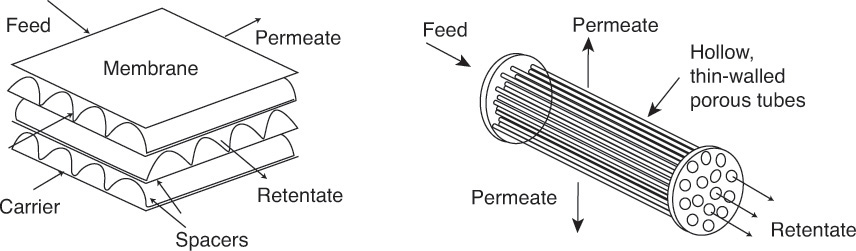
Figure 3.9 Separation using membranes.
Source: Wankat, P. C., Separation Process Engineering, Third Edition, Prentice Hall, Upper Saddle River, New Jersey, 2012.
The schematic on the left shows a crossflow unit where the component transferring across the membrane is swept from the system using a carrier gas. The components not diffusing across exit the unit as retentate. The schematic on the right in Figure 3.9 shows a hollow fiber membrane unit, where permeate diffuses across the tube walls from the feed-retentate stream flowing inside the tubes. Important applications of membrane separations include oxygen-nitrogen separation and desalination.
Several other important separation processes—ion exchange, electrochemical separations, crystallization, and so on—are also studied in this course. The generalized concepts covered in the course provide the foundation for a chemical engineer to design and operate a separation scheme for any mixture.
3.3.1.3 Transport Phenomena
Almost all chemical engineering curricula feature a sequence of two (if on a semester system) or three (if on a quarter system) courses involving the term transport in the course name. These courses are typically offered in the junior (or third) year of the program and are termed Transport and Rate Processes at the University of Idaho. Transport Phenomena is a common name used in many other institutions. The three transport phenomena covered in these courses are momentum transport, energy transport, and mass transport [9].
These courses lay the theoretical foundation for understanding the processes occurring in chemical systems. All processes, whether they involve a simple fluid flow, transfer of heat, or a component, occur at the molecular level. The mechanisms of transfer of the quantities involved—momentum, energy, and mass—are analogous to each other. The theoretical analysis and quantitative results obtained from examining the molecular-level processes for momentum transfer can be extended and applied to the other two transport phenomena.
These courses typically begin with the mathematical description of phenomena associated with the flow of fluids. The similarity between fluid flow behavior and energy/mass transfer is used to extend the mathematical model of momentum transport to energy and mass transport. This quantitative analysis provides the key to understanding the factors that govern the rate of transfer of quantities of interest.
The topics covered in momentum transport include a fundamental description of viscosity and shear forces in a fluid, quantitation of turbulence and frictional losses, and energy balances in fluid systems. Processes occurring at the boundary of a fluid are examined from a microscopic or molecular viewpoint. Energy transport and mass transport build on these concepts to link the heat and mass transfer to the molecular processes in fluids. Figure 3.10 provides a broad overview of the transport phenomena topics and some important concepts.

Figure 3.10 Overview of transport phenomena.
The system behavior at the macroscopic level can always be linked to measurable parameters, such as temperature, flow rate, and so on, using a trial-and-error procedure. Empirical relationships obtained through such an exercise have a limited utility and validity for specific situations. Study of transport phenomena occurring at the microscopic level provides a theoretical basis for explaining the system behavior at the macroscopic level. Relationships between observed quantities and system parameters developed on the basis of this theoretical analysis are scientifically valid and have general applicability to other systems. Transport phenomena courses thus equip a student with the knowledge to analyze any situation based on sound science. This knowledge imparts an ability to predict accurately the system response to changes in operating parameters and hence an ability to increase the efficiency of operations, whether it is separations, heat transfer, or simply material flow. The quantitative tools acquired by the student are used in the design of heat transfer, fluid flow, and separations equipment.
3.3.1.4 Process Control
Most process plants operate in continuous, steady-state mode. Maintaining the plant conditions at the specified design values is essential for meeting the product specifications. At the same time, no processing step is immune from experiencing disturbances due to variations in the quality of raw material streams, malfunctioning of units (pump failure, loss of electrical power, etc.), inlet flow rate fluctuations, and many other factors. A chemical engineer must understand the impact of such disturbances on the output of the process and be able to devise and implement corrective measures to control the process within the accepted tolerance of the setpoint of the process. The course on process control is aimed at equipping students with the knowledge of principles and techniques of process control.
Controlling a process requires understanding the stimulus-response behavior of the process unit. The student learns to develop transfer functions—quantitative descriptions of the change in the process output due to a change in input conditions—for the process unit. Controlling the process requires modification of this transfer function by addition of a control element in the process. This control element enables the plant operator to manipulate a variable to maintain the controlled variable at the desired operation point. This principle of process control is explained qualitatively through the following example.
Consider a holding tank that has a continuous inflow and an outflow of a liquid stream. It is required to control the liquid level in the tank at a set value. It is clear that any variation in the inlet stream flow rate, if not controlled, will cause the liquid level to change. When the inlet flow increases, the control strategy should result in increased outflow. The opposite must occur when the inlet flow decreases. This can be accomplished by installing in the outlet line a valve that can be opened or closed on the basis of the disturbance. The tank level is the controlled variable, the desired level is the setpoint, and the outlet flow (more accurately, the valve position) is the manipulated variable. The control scheme is shown in Figure 3.11 [10].
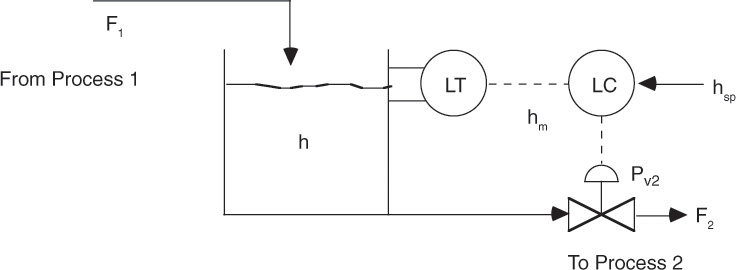
Figure 3.11 Controlling liquid level in a process tank.
Source: Bequette, B. W., Process Control: Modeling, Design and Simulation, Prentice Hall, Upper Saddle River, New Jersey, 2003.
The transfer function for the process describing the relationship between the controlled variable and the feed (inlet flow) is developed, and the desired change in the manipulated variable is quantified on the basis of this transfer function. The process is monitored by making certain measurements. The measured variable may or may not be the same as the controlled variable. In this case, the liquid level or inlet flow rate may serve as the measured variable. It can be seen that several alternative strategies are possible for controlling the liquid level. It may be possible to manipulate the inlet flow rate by installing a control valve in the inlet line, installing an overflow weir, or adding a make-up stream to the system. The process unit downstream of the holding tank may require a set flow to operate, and in this case, the outlet flow rate cannot be a manipulated variable, so an alternative strategy needs to be devised. Temperature, pressure, and concentration are among the other variables that are frequently controlled in chemical processes. Figure 3.12 shows a control scheme for maintaining the temperature of a reactor at the desired setpoint by manipulating the flow rate of the heat transfer medium in the reactor jacket.
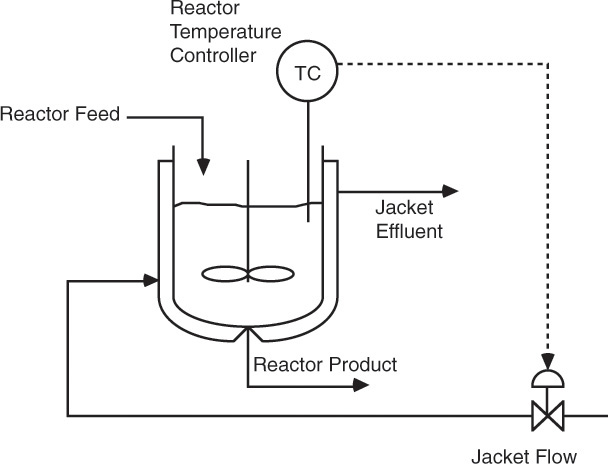
Figure 3.12 Temperature control for a reactor.
Source: Bequette, B. W., Process Control: Modeling, Design and Simulation, Prentice Hall, Upper Saddle River, New Jersey, 2003.
In all cases, the approach for process control is the same: develop the transfer function to quantify the relationships, and design an algorithm to change the manipulated variable to counteract the disturbance. Controlling batch processes involves additional complexity of time dependency. Here the setpoint (the desired value of the controlled variable) may change with time, unlike the continuous processes. Despite this complication, a control algorithm and the control strategy can be developed along the same lines.
3.3.1.5 Elective Courses
Apart from the essential courses, a chemical engineering student will typically have anywhere from three to five technical courses in chemical engineering and related areas as electives. The course offerings vary among institutions depending on the faculty expertise and interests. Materials related courses, such as courses in corrosion, polymer engineering, and ceramic materials, are common if the chemical engineering program is closely aligned with material science and engineering. Several chemical engineering departments have expertise in bio-related areas, and biochemical engineering, bioenergy, and biomedical courses are the available electives. Students may also take as electives relevant courses in environmental engineering, petroleum engineering, semiconductors, and nuclear engineering.
Engineering activities related to the biological or life-sciences area have grown exponentially in recent times, as was seen from the graph of engineering graduates in Chapter 1. A large number of chemical engineering departments have responded to this trend by incorporating relevant courses in their programs. Many curricula mandate a bioengineering course offered within the department or a biology, microbiology, molecular biology, or biochemistry course from other departments and programs, while several others strongly suggest such a course. This trend is reflected both in the names of many chemical engineering departments across the United States, which are now called chemical and biochemical engineering departments (or a variation thereof), and in the emphasis on life-science aspects of chemical engineering in the American Institute of Chemical Engineers (AIChE) definition stated in Chapter 1.
3.3.1.6 Process Design
As mentioned earlier, a capstone design project in the final year of the program completes the formal education of a chemical engineer. This is accomplished at the University of Idaho through a two-semester sequence of courses called Process Analysis and Design I and II. Typically, a student is assigned a comprehensive project that requires him/her to apply the concepts learned in other courses and present a detailed solution. The student invariably is required to submit a technical document and make a presentation to the class, college, or a wider audience. The project may be an individual or group assignment and may involve building working prototypes and conducting demonstration experiments. The scope of the project may be as wide as designing a complete plant for the production of a chemical, or it may be very specific and focused, such as removing a particular contaminant from a specific effluent stream.
Students typically learn the fundamentals of process design and plant economics in the initial part of the course at the beginning of the final year. Cost estimation of process units, accounting for capital and operating costs, cash flow, and return on investment analysis are some to the topics covered in the course [11]. Students learn the techniques to assign economic value to the engineering activities. Students may also get experience working with commercial process simulation software used in industry. The design course is thus the final step in preparing a student for a chemical engineering career. It is not unusual for a private industry or a public sector employer of engineers to agree to provide actual engineering problems related to their businesses as senior design problems for student groups. Such entities may participate in the design process by providing funding or advice as appropriate. Such real-world problems are often especially beneficial to the learning process.
Students enrolling in these advanced courses should have the necessary academic preparation, which is accomplished through the fundamental chemical engineering and engineering science courses.
3.3.2 Fundamental Chemical Engineering Courses
The foundation for the advanced chemical engineering courses is laid in the second and third (sophomore and junior) years of the program through two fundamental chemical engineering courses: Material and Energy Balance and Chemical Engineering Thermodynamics. Both these courses are described in the following sections.
3.3.2.1 Material and Energy Balance
Scheduled in the second year of the program, this course is possibly the most important course in the chemical engineering curriculum. The title of course is generally Material and Energy Balance, but it may sometimes be called Chemical Engineering Principles, Introduction to Chemical Processes, or a variation thereof. Simply put, the course is aimed at teaching the students the techniques to conduct the material and energy audits on the process. Material balance involves accounting for quantities of materials flowing in and out of a process unit, a sequence of units, and the entire plant. Energy balance is, similarly, the quantification of energy flows in and out of the system for which this analysis is conducted. In essence, the course teaches students how to apply the principles of conservation of mass and conservation of energy to units in chemical processes. These units may involve reacting or nonreacting systems, as well as single-phase and multiphase systems. Figure 3.13 presents an overview of this course.

Figure 3.13 Overview of the Material and Energy Balance course.
Typically, the course starts with the concept of material balances and application of this concept to a single step in a simple, nonreacting system. Subsequent topics include complex combinations of nonreacting units, a single unit involving reaction(s), a combination of reacting and nonreacting units, energy effects not involving reaction, and energy effects in reacting systems [12].
A student, after successful completion of the course, should be able to determine accurately the mass, volume, temperature, and composition of each material stream flowing into and out of a process unit. The process occurring within this unit may be simple mixing, a physical separation, or a complex reaction. The process streams may be solid, liquid, gaseous, or any combination thereof. Similarly, the student should be able to determine accurately the energy flows into and out of the units.
Quantifying the material and energy streams connected to a process unit is a prerequisite to designing that unit. These design principles and techniques are taught in the advanced chemical engineering courses previously described. It is clear that unless the student acquires competence in tackling the material and energy balances, he/she will not be in a position to learn the advanced topics in chemical engineering. The Material and Energy Balance course provides an indicator of the ability of a student to be a chemical engineer, often serving as a “gate” for continuation in the chemical engineering program.
3.3.2.2 Chemical Engineering Thermodynamics
A course in engineering thermodynamics is required of students in all engineering disciplines. In addition, chemical engineering students take, generally in the first semester of the third (junior) year, a specialized thermodynamics course called Chemical Engineering Thermodynamics. Thermodynamics is the engineering science that deals with the interconversion between work and energy. The concept of equilibrium is integral to thermodynamics, and the essence of Chemical Engineering Thermodynamics is the study of equilibrium phenomena in chemical systems. Equilibrium can be understood to be the state of a system from which no change is expected; that is, the system is in a stable state. It follows that any system not at equilibrium will have the tendency to move toward equilibrium. The driving force for any process is the departure from equilibrium.
Chemical Engineering Thermodynamics helps students define equilibrium in terms of thermodynamic quantities, such as enthalpy, entropy, free energy, and so on, and relate these thermodynamic quantities to measurable system properties, such as pressure, temperature, volume, and composition [13]. Students learn about the changes in thermodynamic quantities associated with various chemical processes and how to quantify these changes from the volumetric behavior of substances. Volumetric behavior refers to the pressure-volume-temperature relationship for the substance. The ideal gas law is the simplest equation describing the volumetric behavior. Most substances, as they are not ideal gases, require more complex equations, and students learn about nonideal behavior and equations that describe this behavior.
Students learn to apply the laws of thermodynamics to chemical systems—pure substances and mixtures, single phase and multiphase, reacting and nonreacting. Figure 3.14 provides an overview of the course.

Figure 3.14 Overview of the Chemical Engineering Thermodynamics course.
Concepts learned in Chemical Engineering Thermodynamics are applied in the advanced courses, particularly separation processes and kinetics, and provide the theoretical foundation for process and equipment design.
3.3.3 Engineering Science Courses
The second (sophomore) year of the curriculum also features the engineering science courses that are common to nearly all engineering disciplines. Principles and concepts learned in these courses serve as prerequisites for higher-level engineering courses. A chemical engineering student also gains an exposure to other engineering disciplines and is in a position to interface with civil, electrical, and mechanical engineers as he/she would invariably be required to do throughout his/her professional career.
3.3.3.1 Fluid Mechanics
Piping is the most ubiquitous feature of a chemical plant. Chemical processes typically involve large material flows, and it is essential for a student to understand the physical phenomena occurring in fluid systems. The Fluid Mechanics course teaches the students the application of conservation of mass and energy principles to fluid systems. The two components of Fluid Mechanics are fluid statics, the phenomena associated with fluids at rest, and fluid dynamics, the phenomena associated with fluids in motion. Fluid systems are analyzed at a macroscopic level; that is, in terms of observable bulk properties of the material. The energy balances generally focus on the mechanical energy—kinetic and potential—of the system, with thermal energy contributions playing an insignificant role.
Students gain an understanding of forces acting on fluid elements in stationary and moving fluids. This understanding is useful in determining the forces and pressure in stationary fluids as well as energy and power requirements in flow systems. Substantial costs are incurred in chemical processes in simply moving material from one process unit to another. Figure 3.15 provides an overview of the course.

Figure 3.15 Overview of the Fluid Mechanics course.
The concepts learned in Fluid Mechanics are prerequisite for the transport phenomena courses, which analyze the systems from a microscopic or molecular level. The Fluid Mechanics course helps the students understand the macroscopic behavior of the fluids. The transport phenomena courses delve into the explanation of this observed behavior through the analysis of microscopic processes occurring in the fluid.
3.3.3.2 Engineering Thermodynamics
As mentioned previously, thermodynamics is the branch of science that deals with the interconversion of energy and work. Engineers, at a very basic level, are individuals who deal with engines—machinery and processes—to obtain useful work for the benefit of the society. This work is obtained at the expense of energy, and engineers need to have fundamental understanding of the concepts that govern the relationship between energy and work. Engineering Thermodynamics teaches students the laws of thermodynamics and applications of these laws to various systems. Students also learn about the thermodynamic quantities (enthalpy, entropy, free energy, etc.), as mentioned earlier, and the changes in these quantities in various types of processes. The process may be isothermal (occurring at constant temperature), isobaric (at constant pressure), or isochoric (at constant volume). Engineers construct power conversion cycles consisting of combinations of these and other processes. These concepts are then applied to the analysis of energy or power conversion cycles that involve a cyclic sequence of different processes. The course also covers macroscopic analysis of heat transport processes. An overview of the Engineering Thermodynamics course is shown in Figure 3.16.

Figure 3.16 Overview of the Engineering Thermodynamics course.
The concepts covered in Engineering Thermodynamics are prerequisites for courses such as Chemical Engineering Thermodynamics and Transport Phenomena. It can be seen that the topics covered in the Engineering Thermodynamics course are more general in nature than those in the Chemical Engineering Thermodynamics course. Although some chemical engineering programs conform to this arrangement of the two courses, several other programs may simply have two chemical engineering thermodynamics courses, with the topical areas corresponding roughly to those described previously. If the first engineering thermodynamics course is taught by the faculty in the chemical engineering program with the enrollment restricted primarily to the chemical engineering majors, then typically it is named Chemical Engineering Thermodynamics I. If the course is offered by any faculty member in the college of engineering, with enrollment open to any engineering student, it typically appears as Engineering Thermodynamics. Regardless of the specific arrangement, the two courses function to provide discussion of fundamental concepts of engineering thermodynamics and their application to chemical engineering systems.
3.3.3.3 Engineering Statics and Electrical Circuits
Chemical engineering students also typically are exposed to the concepts related to civil engineering and electrical engineering through courses such as Engineering Statics and Electrical Circuits. The students learn about force balance on rigid bodies, equilibrium, strength of materials and structures, and so on, in the Engineering Statics course. Students learn how to analyze trusses and frames and other load-bearing structures. This knowledge is useful in the design of chemical process equipment, where the chemical engineer must take into consideration the load demand an equipment will place on the supporting structure.
Similarly, the Electrical Circuits course exposes the chemical engineering students to concepts related to electrical systems. Students learn to analyze the steady-state and transient behavior of electrical energy and power systems. This knowledge is also useful in the design of processes and equipment where the chemical engineer must take into account electrical requirements imposed by the unit.
3.3.3.4 Computer Programming
Most chemical engineering programs require students to take a course in computer science, typically a course in a programming language. Historically, most engineers studied FORTRAN programming, but the emphasis has shifted to other newer languages and software in recent years. Students generally take a course in programming languages such as Visual Basic, C/C++, MATLAB, or other software languages. Irrespective of the specific language, students learn about algorithm development and code formulation in the course. These skills are invaluable for any engineer, as growing computational power enables an engineer to tackle problems of increasing complexity.
Success in these fundamental courses, in turn, is based on basic science and mathematics courses, scheduled in the first two years of the curriculum. These courses are described next.
3.3.4 Fundamental Science and Mathematics Courses
Chemical engineering students take several chemistry, physics, and mathematics courses during the first (freshman) and second (sophomore) years.
3.3.4.1 Chemistry Courses
Most engineering students in other disciplines take perhaps one course in chemistry, typically in the first semester of the first (freshman) year. Chemical engineering students take a significantly larger number of chemistry courses. The courses in the first year of study expose students to concepts in general and inorganic chemistry. The next two to three semesters involve instruction in organic chemistry and physical chemistry. After completion of these courses a student should have knowledge of the following areas:
• Kinetics and thermodynamics
• Equilibrium
• Acids and bases
• Electrochemistry
• Nuclear chemistry
• Synthesis and properties of organic compounds
• Biochemistry
• Quantum mechanics
Each course consists of lecture and laboratory components, giving students an opportunity to gain both conceptual and practical knowledge.
In addition to these required courses, students may take more chemistry courses to satisfy the science elective requirements. Chemical engineering students in many institutions may be able to work these additional chemistry courses into their study plans to satisfy requirements for the bachelor’s degree in chemistry.
3.3.4.2 Physics Courses
Chemical engineering students typically take two physics courses, generally completed by the third semester. The topics covered in these courses generally include the following:
• Kinematics and dynamics
• Newton’s laws
• Friction
• Static equilibrium
• Work and energy
• Gravity and central forces
• Momentum
• Electrical fields and potentials
• Magnetism
• AC/DC circuits, capacitance, and inductance
As with chemistry courses, these courses also have lecture and laboratory components.
3.3.4.3 Mathematics Courses
Chemical engineering students, similar to other engineering students, typically take a sequence of three semester courses in mathematics covering various topics in analytical geometry and calculus. The topics covered in these courses include functions, limits and continuity, series, integration and differentiation, vectors, algebraic and transcendental equations, numerical techniques, conics and solid geometry, and so on. These courses are generally prerequisites for a course on differential equations, which is one of the most important courses in the education of an engineer. Students learn about first- and higher-order differential equations; initial and boundary value problems; solution techniques for equations, including series solutions and Laplace transforms; and systems of linear equations. These differential equations are representative of the mathematical models of systems encountered in the engineering field.
The relationship between various science and engineering courses that are essential for the education of a chemical engineer is summarized in Figure 3.17.

Figure 3.17 Hierarchy of technical courses in chemical engineering curriculum.
3.3.5 General Education Courses
In addition to these technical courses, a proper number of general education, humanities, and social science courses is absolutely essential to complete the education of a chemical engineer. Two of these courses play almost as important a role in an individual’s career as any of the technical courses: economics and communication.
A course in economics is essential for the engineer to understand the ultimate driving force for most, if not all, professional activities he/she will be involved in. Economic considerations play a major role in evaluation of alternative technologies and processes and often dictate the ultimate choice. The course provides an engineer with not only an understanding of the interplay of economics and technology but also a perspective about the role of a chemical engineer in a societal context.
Competence in reaction engineering, thermodynamics, transport phenomena, and separations is viewed as an essential qualification by the industry. However, an ability to communicate is an equally important skill that an employer looks for in a prospective employee [14]. Chemical engineers need to interact and communicate with a broad range of people having varying levels of technical knowledge. They need to communicate with chemical and other engineers, technical managers and administrators, sales/marketing and finance personnel, and plant operators and technicians, among others. The chemical engineer should be able to convey his/her ideas clearly and succinctly to all these people. A chemical engineer will quickly discover that technically sound, logical ideas that benefit the process need to be articulated properly even in a peer group of chemical engineers for them to accept it. A course that helps develop written and oral communication skills is a must for a budding chemical engineer.
Economics and communication are probably the two most important general education courses, but the value of the other general education courses cannot be underestimated. The humanities and social science courses in disciplines such as philosophy, history, sociology, and so on are absolutely essential for the holistic education of an individual. These courses provide a moral and ethical framework for one’s thoughts and actions, present the context for the development of societies, and promote an understanding and appreciation of the diversity of viewpoints, and they provide many other such benefits that can only lead to a better chemical engineer.

Leave a Reply