Now that you have had a chance to review the ideal gas law applied to simple problems, let’s apply the ideal gas law in material balances. The only difference between the subject matter of Chapters 3 through 6 and this chapter is that here the amount of material can be specified in terms of p, V, and T rather than solely as mass or moles. For example, the basis for a problem, or the quantity to be solved for, might be a volume of gas at a given temperature and pressure rather than a mass of gas. The next two examples illustrate balances for problems similar to those you have encountered before, but now involving gases.
Example 7.4. Material Balances for a Process Involving Combustion
To evaluate the use of renewable resources, an experiment was carried out with rice hulls. After pyrolysis, the product gas analyzed 6.4% CO2, 0.1% O2, 39% CO, 51.8% H2, 0.6% CH4, and 2.1% N2. It entered a combustion chamber at 90°F and a pressure of 35.0 in. Hg and was burned with 40% excess air (dry) at 70°F and an atmospheric pressure of 29.4 in. Hg; 10% of the CO remains. How many cubic feet of air were supplied per cubic foot of entering gas? How many cubic feet of product gas were produced per cubic foot of entering gas if the exit gas was at 29.4 in. Hg and 400°F?
Solution
This is an open, steady-state system with reaction. The system is the combustion chamber.
Steps 1–4
Figure E7.4 illustrates the process and notation. With 40% excess air, certainly all of the CO, H2, and CH4 should burn to CO2 and H2O; apparently, for some unknown reason, not all of the CO burns to CO2. No CH4 or H2 appears in the product gas. The components of the product gas are shown in the figure.
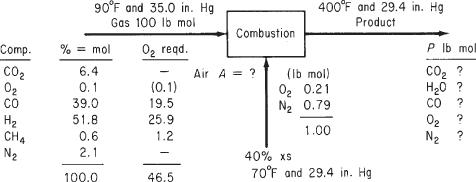
Figure E7.4
Step 5
You could take 1 ft3 at 90°F and 35.0 in. Hg as the basis and convert the volume to moles, but it is just as easy to take 100 lb (or kg) mol as a basis because then % = lb (or kg) mol. Because only ratios of volumes are asked for, not absolute amounts, at the end of the problem you can convert pound (or kilogram) moles to cubic feet.
Basis: 100 lb mol of pyrolysis gas
Step 4 (continued)
The entering air can be calculated from the specified 40% excess air; the reactions for complete combustion are
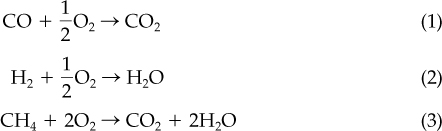
The moles of oxygen required are listed in Figure E7.4. (We will omit the units—pound moles—in what follows.) The excess oxygen is
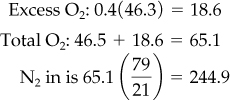
Total moles of air in are 244.9 + 65.1 = 310.0 lb mol.
Finally, use the specification for CO to assign nco = 0.10(39.0) = 3.9.
Steps 6 and 7
Degree-of-freedom analysis:
Unknowns (5): nCO2, nO2, nN2, nH2O, P
Equations (5):
Element balances (4): C, H, O, N
Implicit equations (1): P = ∑ni
Steps 8 and 9
Make the element balances in moles to calculate the unknown quantities, and substitute the value of 3.9 for the number of moles of CO exiting.

The solutions of these equations are
nN2 = 247 nCO2 = 42.1 nH2O = 53.0 nO2 = 20.55
The total moles exiting calculated from the implicit equation sum to 366.6 mol.
Finally, you can convert the pound moles of air and products that were calculated on the basis of 100 lb mol of pyrolysis gas to the volumes of gases at the states requested using the ideal gas law:
Tgas = 90 + 460 = 550°R → 306 K
Tair = 70 + 460 = 530°R → 294 K
Tproduct = 400 + 460 = 860°R → 478 K
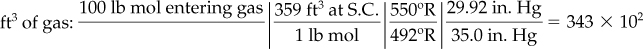
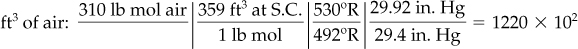
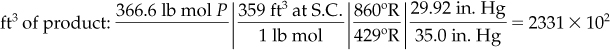
The answers to the questions are

Example 7.5. Material Balance without Reaction
Gas at 15°C and 105 kPa is flowing through an irregular duct. To determine the rate of flow of the gas, CO2 from a tank is steadily passed into the gas stream. The flowing gas, just before mixing with the CO2, analyzes 1.2% CO2 by volume. Downstream, after mixing, the flowing gas analyzes 3.4% CO2 by volume. As the CO2 that was injected exited the tank, it was passed through a rotameter and found to flow at the rate of 0.0917 m3/min at 7°C and 131 kPa. What was the rate of flow of the entering gas in the duct in cubic meters per minute?
Solution
This is an open, steady-state system without reaction. The system is the duct. Figure E7.5 is a sketch of the process.
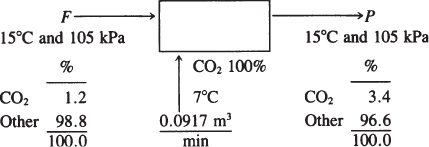
Figure E7.5
Steps 1–4
The data are presented in Figure E7.5.
Both F and P are at the same temperature and pressure.
Step 5
Should you take as a basis 1 min → 0.0917 m3 of CO2 at 7°C and 131 kPa? The gas analysis is in volume percent, which is the same as mole percent. We could convert all of the gas volumes to moles and solve the problem in terms of moles, but there is no need to do so because we can just as easily convert the known flow rate of the addition of CO2 to 15°C and 105 kPa and solve the problem using cubic meters for each stream since all of the streams will be at the same conditions.
Steps 6 and 7
The unknowns are F and P, and you can make two independent component balances, CO2 and “other”; hence, the problem has zero degrees of freedom.
Steps 7–9
The “other” balance (in cubic meters at 15°C and 105 kPa) is

The CO2 balance (in cubic meters at 15°C and 105 kPa) is
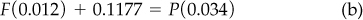
The total balance (in cubic meters at 15°C and 105 kPa) is

Note that the “other” is a tie component. Select Equations (a) and (c) to solve. The solution of Equations (a) and (c) gives
F = 5.17 m3/min at 15°C and 105 kPa
Step 10 (check)
Use the redundant Equation (b):
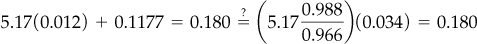
The equation checks out to a satisfactory degree of precision.
Self-Assessment Test
Questions
1. What are the dimensions of T, P, V, n, and R?
2. List the standard conditions for a gas in the SI and American Engineering systems of units.
3. How do you calculate the density of an ideal gas at S.C.?
4. Can you use the respective specific molar densities (mole/volume) of the gas and the reference gas to calculate the specific gravity of a gas?
5. A partial pressure of oxygen in the lungs of 100 mm Hg is adequate to maintain oxygen saturation of the blood in a human. Is this value higher or lower than the partial pressure of oxygen in the air at sea level?
6. An exposure to a partial pressure of N2 of 1200 mm Hg in air has been found by experience not to cause the symptoms of N2 intoxication to appear. Will a diver at 60 m be affected by the N2 in the air being breathed?
Problems
1. Calculate the volume in cubic feet of 10 lb mol of an ideal gas at 68°F and 30 psia.
2. A steel cylinder of volume 2 m3 contains methane gas (CH4) at 50°C and 250 kPa absolute. How many kilograms of methane are in the cylinder?
3. What is the value of the ideal gas constant R to use if the pressure is to be expressed in atmospheres, the temperature in kelvin, the volume in cubic feet, and the quantity of material in pound moles?
4. Twenty-two kilograms per hour of CH4 are flowing in a gas pipeline at 30°C and 920 mm Hg. What is the volumetric flow rate of the CH4 in cubic meters per hour?
5. A gas has the following composition at 120°F and 13.8 psia:

a. What is the partial pressure of each component?
b. What is the volume fraction of each component?
6. A furnace is fired with 1000 ft3/hr at 60°F and 1 atm of a natural gas having the following volumetric analysis: CH4 80%, C2H6 16%, O2 2%, CO2 1%, and N2 1%. The exit flue gas temperature is 800°F and the pressure is 760 mm Hg absolute; 15% excess air is used and combustion is complete. Calculate (a) the volume of CO2 produced per hour; (b) the volume of H2O vapor produced per hour; (c) the volume of N2 produced per hour; (d) the total volume of flue gas produced per hour.
Thought Problems
1. In a test of the flow of gases through a pipe, pure hydrogen was found to flow at a volumetric flow 22 times that of carbon dioxide. When the hydrogen was diluted with carbon dioxide entering the pipe midway from the ends, the exit flow rate was less than that of pure hydrogen. Explain the observed differences.
2. A pair of identical balloons are inflated with air to the same pressure and tied to a stick that is held in the center by a string. The balloons are the same distance from the center of the stick so that the stick remains horizontal to the ground. When the left-hand balloon is carefully punctured, will the stick rotate down from the left, rotate up from the left, or remain horizontal?
Discussion Question
In a demonstration, a 30-cm-diameter balloon was filled to two-thirds of its maximum pressure with SF6, a gas. Students measured the balloon’s diameter for 10 days, at which time the balloon burst. No one ever touched it. Explain how this could happen. (Note: The balloon was not defective.)

Leave a Reply