From a single-component two-phase system, let’s extend the discussion to a more complicated system, namely, a system with two components in the gas phase together with a single-component liquid system. An example of such a system is water and a noncondensable gas, such as air. The equilibrium relationships for the water and air help explain how rain is formed and lead to a number of meteorological terms, such as the dew point and the humidity of the air. Moreover, the equilibrium relationship for two-component gas/single-component liquid systems is used industrially to describe and design many systems, including cooling towers, in which water is cooled by evaporation, and stripping systems, in which a volatile component is removed from a liquid by contacting the liquid with a noncondensable gas.
7.4.1 Saturation
When any noncondensable gas (or a gaseous mixture) comes in contact with a liquid, the gas will acquire molecules from the liquid. If contact is maintained for a sufficient period of time, vaporization continues until equilibrium is attained, at which time the partial pressure of the vapor in the gas will equal the vapor pressure of the liquid at the temperature of the system. Regardless of the duration of contact between the liquid and gas, after equilibrium is reached, no more net liquid will vaporize into the gas phase. The gas is then said to be saturated with the particular vapor at the given temperature. We also say that the gas mixture is at its dew point. The dew point for the mixture of pure vapor and noncondensable gas means the temperature at which the vapor would just start to condense if the temperature were very slightly reduced. At the dew point, the partial pressure of the vapor is equal to the vapor pressure of the volatile liquid.
Consider a gas partially saturated with water vapor at p and T. If the partial pressure of the water vapor is increased by increasing the total pressure on the system, eventually the partial pressure of the water vapor will equal p* at T of the system. Because the partial pressure of water cannot exceed p* at that temperature, a further attempt to increase the pressure will result in water vapor condensing at constant T and p. Thus, p* represents the maximum partial pressure that water can attain at that temperature, T.
Do you have to have liquid present for saturation to occur? Really, no; only a minute drop of liquid at equilibrium with its vapor will suffice.
What use can you make of the information or specification that a noncondensable gas is saturated? Once you know that a gas is saturated, you can determine the composition of the vapor-gas mixture from knowledge of the vapor pressure of the vapor (or the temperature of the saturated mixture) to use in material balances. From Chapter 6 you should recall that the ideal gas law applies to both air and water vapor at atmospheric pressure with excellent precision. Thus, we can say that the following relations hold at saturation:pH2OVpairV=nH2ORTnairRT
(7.5)
orpH2Opair=pH2O*pair=nH2Onair=ptotal−pairpair
(7.6)
because V and T are the same for the air and water vapor.
Also,yH2O=pH2Optotal=pH2Opair+pH2O=1−yair
(7.7)
As a numerical example, suppose you have a saturated gas, say, water in air at 51°C, and the pressure on the system is 750 mm Hg absolute. What is the partial pressure of the air? If the air is saturated, you know that the partial pressure of the water vapor is p* at 51°C. You can use the physical property software on the website that accompanies this text, or use the steam tables, and find that p* = 98 mm Hg. Then
Pair = 750 − 98 = 652 mm Hg
Furthermore, the vapor-air mixture has the following composition:yH2O=pH2Optotal=98750=0.13yair=pairptotal=652750=0.87
Example 7.7 Calculation of the Dew Point of the Products of Combustion
Problem Statement
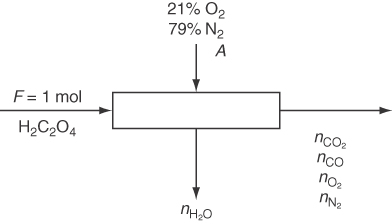
Oxalic acid (H2C2O4) is burned at atmospheric pressure with 4% excess air so that 65% of the carbon burns to CO. Calculate the dew point of the product gas.
Solution
The solution of the problem involves the following steps:
- Calculate the combustion products via material balances.
- Calculate the mole fraction of the water vapor in the combustion products as indicated just prior to this example.
- Assume a total pressure, say, 1 atm, and calculate yH2O ptotal=pH2O in the combustion products. At equilibrium pH2O will be the vapor pressure pH*2O.
- Condensation (at constant total pressure) would be possible when pH2O* equals the calculated pH2O. This value is the dew point.
- Look up the temperature corresponding to pH2O in the saturated steam tables.
Steps 1–5
Basis: 1 mol of H2C2O4
The figure and data are given. The chemical reaction equation for the combustion of oxalic acid is given asH2C2O4+0.5 O2→2 CO2+H2OH2C2O4 → 2 CO+H2O+0.5 O2
Step 4
O2 required:1 mol H2C2O410.5 mo l O21 mol H2C2O4=0.5 mol O2(note oxygen in oxalic acid)
Moles of O2 in with air including excess:
(1 + 0.04) (0.5 mol O2) = 0.52 mol O2
Therefore, 0.52/0.21 = 2.48 mol air enters containing 1.96 mol nN2.
Specifications: 65% of the carbon burns to CO: (0.65) (2) = 1.30.
The results:
| Element Material Balances | ||
|---|---|---|
| Element | In (mol) | Out (mol) |
| C | 2 | nCO2+nCO or 0.70+1.30 |
| H | 2 | 2nH2O |
| N | 1.96 × 2 | 1.96 × 2 |
| O | 0.52 × 2 + 4 | 2nCO2+nCO+nO2 or 2(0.70) + 1.30+ 1.0+2nO2 |
nH2O=1.0; nCO2=0. 7; nCO=1.3; nO2=0.67; nN2=1.96; total mol=5.63yH2O=1 mol H2O/6 .78 mol=0.178
The partial pressure of the water in the product gas (at an assumed atmospheric pressure) determines the dew point of the stack gas; that is, the temperature of saturated steam that equals the partial pressure of the water is equal to the dew point of the product gas:pH2O*=yH2O(ptotal)=0.178 (101.3 kPa)=18 .0 kPa (i.e., 2.61 psia)
From the steam tables, T = 136°F.

Leave a Reply