Among the non-conventional methods to desalinate brackish water or seawater is solar distillation. This process requires a comparatively simple technology and can be operated by unskilled workers. Also, due to the low maintenance requirement, it can be used anywhere with a smaller number of problems.
A representative example of the direct collection system is the typical solar still, which uses the greenhouse effect to evaporate salty water. It consists of a basin in which a constant amount of seawater is enclosed in an inverted V-shaped glass envelope (see Figure 8.1(a)). The sun’s rays pass though the glass roof and are absorbed by the blackened bottom of the basin. As the water is heated, its vapor pressure is increased. The resultant water vapor is condensed on the underside of the roof and runs down into the troughs, which conduct the distilled water to the reservoir. The still acts as a heat trap because the roof is transparent to the incoming sunlight but opaque to the infrared radiation emitted by the hot water (greenhouse effect). The roof encloses the vapor, prevents losses, and keeps the wind from reaching and cooling the salty water. Another basic type of solar still is the single-slope one shown in Figure 8.1(b), also called an asymmetric solar still oriented in a north–south direction.
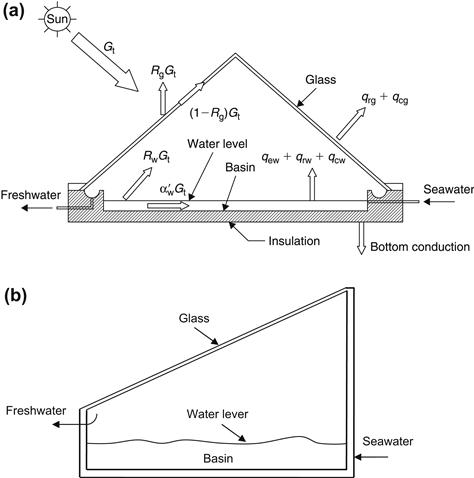
FIGURE 8.1 Schematic diagrams of the basic designs of solar stills. (a) Double-slope solar still showing also the energy flows. (b) Single-slope or asymmetric solar still.
Figure 8.1(a) shows the various components of energy balance and thermal energy loss in a conventional double-slope symmetrical solar distillation unit (also known as a roof-type or greenhouse-type solar still). The still consists of an airtight basin, usually constructed out of concrete galvanized iron sheet, or fiber-reinforced plastic, with a top cover of transparent material such as glass or plastic. The inner surface of the base, known as the basin liner, is blackened to efficiently absorb the solar radiation incident on it. There is also a provision to collect distillate output at the lower ends of the top cover. The brackish or saline water is fed inside the basin for purification using solar energy.
The stills require frequent flushing, which is usually done during the night. Flushing is performed to prevent salt precipitation. Design problems encountered with solar stills are brine depth, vapor tightness of the enclosure, distillate leakage, methods of thermal insulation, and cover slope, shape, and material (Eibling et al., 1971). A typical still efficiency, defined as the ratio of the energy utilized in vaporizing the water in the still to the solar energy incident on the glass cover, is 35% (maximum) and daily still production is about 3–4 l/m2 (Daniels, 1974).
Talbert et al. (1970) gave an excellent historical review of solar distillation. Delyannis and Delyannis (1973) reviewed the major solar distillation plants around the world. This review also includes the work of Delyannis (1965), Delyannis and Piperoglou (1968), and Delyannis and Delyannis (1970). Malik et al. (1982) reviewed the work on passive solar distillation system until 1982, and this was updated up to 1992 by Tiwari (1992), who also included active solar distillation. Kalogirou (1997a) also reviewed various types of solar stills.
Several attempts have been made to use cheaper materials, such as plastics. These are less breakable, lighter in weight for transportation, and easier to set up and mount. Their main disadvantage is their shorter life. Many variations of the basic shapes shown in Figure 8.1 have been developed to increase the production rates of solar stills (Eibling et al., 1971; Tleimat, 1978; Kreider and Kreith, 1981). Some of the most popular are shown in Figure 8.2. Most of these designs also include provisions for rainfall collection.
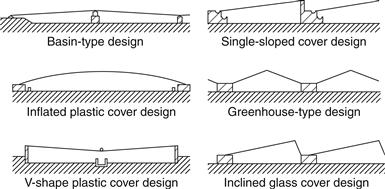
FIGURE 8.2 Common designs of solar stills.
8.3.1 Classification of solar distillation systems
On the basis of various modifications and modes of operation introduced in conventional solar stills, solar distillation systems are classified as passive or active. In active solar stills, an extra thermal energy by external equipment is fed into the basin of a passive solar still for faster evaporation. The external equipment may be a collector–concentrator panel, waste thermal energy from an industrial plant, or a conventional boiler. If no such external equipment is used, then that type of solar still is known as a passive solar still. Types of solar stills available in literature are conventional solar stills, a single-slope solar still with passive condenser, a double-condensing chamber solar still, a vertical solar still (Kiatsiriroat, 1989), a conical solar still (Tleimat and Howe, 1967), an inverted absorber solar still (Suneja and Tiwari, 1999), and a multiple-effect solar still (Adhikari et al., 1995; Tanaka et al., 2000a, b).
Other researchers used different techniques to increase the production of stills. Rajvanshi (1981) used various dyes to enhance performance. These dyes darken the water and increase its solar radiation absorptivity. With the use of black naphthylamine at a concentration of 172.5 ppm, the still output could be increased by as much as 29%. The use of these dyes is safe because evaporation in the still occurs at 60 °C, whereas the boiling point of the dye is 180 °C.
Akinsete and Duru (1979) increased the production of a still by lining its bed with charcoal. The presence of charcoal leads to a marked reduction in start-up time. Capillary action by the charcoal partially immersed in a liquid and its reasonably black color and surface roughness reduce the system thermal inertia.
Lobo and Araujo (1978) developed a two-basin solar still. This still provides a 40–55% increase in the freshwater produced as compared with a standard still, depending on the intensity of solar radiation. The idea is to use two stills, one on top of the other, the top one made completely from glass or plastic and separated into small partitions. Similar results were obtained by Al-Karaghouli and Alnaser (2004a, b), who compared the performance of single- and double-basin solar stills.
Frick and von Sommerfeld (1973), Sodha et al. (1981), and Tiwari (1984) developed a simple multiple-wick-type solar still, in which blackened wet jute cloth forms the liquid surface. Jute cloth pieces of increasing lengths were used, separated by thin black polyethylene sheets resting on foam insulation. Their upper edges were dipped in a saline water tank, where capillary suction provided a thin liquid sheet on the cloth, which was evaporated by solar energy. The results showed a 4% increase in still efficiency above conventional stills.
Evidently the distance of the gap between the evaporator tray and the condensing surface (glass cover) has a considerable influence on the performance of a solar still that increases with decreasing gap distance. This led to the development of a different category of solar stills, the cascaded-type solar still (Satcunanathan and Hanses, 1973). This consists mainly of shallow pools of water arranged in a cascade, as shown in Figure 8.3, covered by a slopping transparent enclosure. The evaporator tray is usually made of a piece of corrugated aluminum sheet (similar to the one used for roofing) painted flat black.
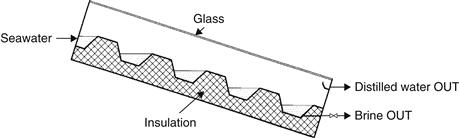
FIGURE 8.3 Schematic of a cascaded solar still.
Thermodynamic and economic analysis of solar stills is given by Goosen et al. (2000). Boeher (1989) reported on a high-efficiency water distillation of humid air with heat recovery, with a capacity range of 2–20 m3/day. Solar still designs in which the evaporation and condensing zones are separated are described in Hussain and Rahim (2001) and El-Bahi and Inan (1999). In addition, a device that uses a “capillary film distiller” was implemented by Bouchekima et al. (2001) and a solar still integrated in a greenhouse roof was reported by Chaibi (2000). Active solar stills in which the distillation temperature is increased by flat-plate collectors connected to the stills are described by Kumar and Tiwari (1998), Sodha and Adhikari (1990), and Voropoulos et al. (2001).
8.3.2 Performance of solar stills
Solar stills are the most widely analyzed desalination systems. The performance of a conventional solar distillation system can be predicted by various methods, such as computer simulation, periodic and transient analysis, iteration methods, and numerical methods. In most of these methods, the basic internal heat and mass transfer relations, given by Dunkle (1961), are used.
Dunkle’s (1961) procedure is summarized by Tiwari et al. (2003). According to this procedure, the hourly evaporation per square meter from a solar still is given by:
![]() (8.26)
(8.26)
Pw = partial vapor pressure at water temperature (N/m2).
Pg = partial vapor pressure at glass temperature (N/m2).
hcw = convective heat transfer coefficient from water surface to glass (W/m2 °C).
The partial vapor pressures at the water and glass temperatures can be obtained from Eq. (5.21). The convective heat transfer coefficient can be obtained from:
![]() (8.27)
(8.27)
d = average spacing between water and glass surfaces (m).
k = thermal conductivity of humid air (W/m °C).
Gr = Grashof number (dimensionless).
Pr = Prandl number (dimensionless).
The dimensionless quantities are given by:
![]() (8.28)
(8.28)
![]() (8.29)
(8.29)
g = gravitational constant = 9.81 m/s2.
β = coefficient of volumetric expansion of fluid (1/K).
ΔT = temperature difference between surface and fluid (K).
μ = dynamic viscosity of fluid (kg/m s).
v = kinetic viscosity of fluid (m2/s).
cp = specific heat of fluid (J/kg K).
By using Eqs (8.26) and (8.27), the hourly distillate output per square meter from a distiller unit ![]() is given by:
is given by:
![]() (8.30)
(8.30)
![]() (8.31)
(8.31)
where
![]() (8.32)
(8.32)
Lv = latent heat of vaporization (kJ/kg)
It should be noted that, in the preceding equation, the product GrPr is known as the Rayleigh number, Ra. The constants C and n are calculated by regression analysis for known hourly distillate output (Dunkle, 1961), water and condensing cover temperatures, and design parameters for any shape and size of solar stills (Kumar and Tiwari, 1996).
According to Tiwari (2002), the instantaneous efficiency of a distiller unit is given as:
![]() (8.33)
(8.33)
Simplifying this equation, we can write:
![]() (8.34)
(8.34)
Tw0 = temperature of basin water at t = 0 (°C).
The preceding equation describes the characteristic curve of a solar still in terms of the solar still efficiency factor (F′), effective transmittance–absorptance product ![]() and overall heat loss coefficient (UL) (Tiwari and Noor, 1996).
and overall heat loss coefficient (UL) (Tiwari and Noor, 1996).
A detailed analysis of the equations of ηi justifies that the overall top loss coefficient (UL) should be maximum for faster evaporation, which results in higher distillate output.
The meteorological parameters—wind velocity, solar radiation, sky temperature, ambient temperature—salt concentration, algae formation on water, and mineral layers on the basin liner affect significantly the performance of solar stills (Garg and Mann, 1976). For better performance of a conventional solar still, the following modifications were suggested by various researchers:
• Reducing the bottom loss coefficient.
• Reducing the water depth in a basin–multiwick solar still.
• Using internal and external condensers.
• Using the back wall with cotton cloth.
• Using an energy storage element.
• Using a multiwick solar still.
• Using an inclined solar still.
• Increasing the evaporative area.
About a 10–15% change in the overall daily yield of solar stills due to variations in climatic and operational parameters within the expected range has been observed.
EXAMPLE 8.3
A solar still has water and glass temperatures equal to 55 °C and 45 °C, respectively. The constants C and n are determined experimentally and found to be C = 0.032 and n = 0.41. If the convective heat transfer coefficient from water surface to glass is 2.48 W/m2 K, estimate the hourly distillate output per square meter from the solar still.
Solution
From Eq. (5.21) and the temperatures of the water and glass, the partial pressures can be obtained as:
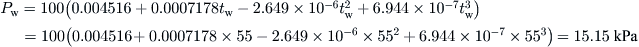
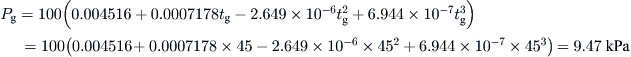
From Eq. (8.26),
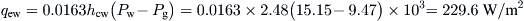
From steam tables, the latent heat of vaporization at 55 °C (water temperature) is 2370.1 kJ/kg.
From Eq. (8.30),
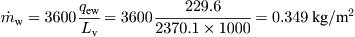
8.3.3 General comments
Generally, the cost of water produced in solar distillation systems depends on the total capital investment to build the plant, the maintenance requirements, and the amount of water produced. No energy is required to operate the solar stills unless pumps are used to transfer the water from the sea. Therefore, the major share of the water cost in solar distillation is that of amortization of the capital cost. The production rate is proportional to the area of the solar still, which means that the cost per unit of water produced is nearly the same regardless of the size of the installation. This is in contrast with conditions for freshwater supplies as well as for most other desalination methods, where the capital cost of equipment per unit of capacity decreases as the capacity increases. This means that solar distillation may be more attractive than other methods for plants of small sizes. Howe and Tleimat (1974) reported that the solar distillation plants with capacity of less than 200 m3/day are more economical than other plants.
Kudish and Gale (1986) presented the economic analysis of a solar distillation plant in Israel, assuming the maintenance cost of the system to be constant. An economic analysis for basin and multiple-wick solar stills was carried out by various scientists (Delyannis and Delyannis, 1985; Tiwari and Yadav, 1985; Mukherjee and Tiwari, 1986). Their economic analyses incorporated the effects of subsidy, rainfall collection, salvage value, and maintenance cost of the system.
Zein and Al-Dallal (1984) performed chemical analysis to find out its possible use as potable water and results were compared with tap water. They concluded that the condensed water can be mixed with well water to produce potable water and the quality of this water is comparable with that obtained from industrial distillation plants. The tests performed also showed that impurities such as nitrates, chlorides, iron, and dissolved solids in the water are completely removed by the solar still.
Although the yield of solar stills is very low, their use may prove to be economically viable if small water quantities are required and the cost of pipework and other equipment required to supply an arid area with naturally produced freshwater is high.
Solar stills can be used as desalinators for remote settlements where salty water is the only water available, power is scarce, and demand is less than 200 m3/day (Howe and Tleimat, 1974). This is very feasible if setting of water pipelines for such areas is uneconomical and delivery by truck is unreliable or expensive. Since other desalination plants are uneconomical for low-capacity freshwater demand, solar stills are viewed as means for communities to attain self-reliance and ensure a regular supply of freshwater.
In conclusion, solar stills are the cheapest, with respect to their initial cost, of all available desalination systems in use today. They are direct collection systems, which are very easy to construct and operate. The disadvantage of solar stills is the very low yield, which implies that large areas of flat ground are required. It is questionable whether solar stills can be viable unless cheap desert-like land is available near the sea. However, obtaining freshwater from saline or brackish water with solar stills is useful for arid remote areas where no other economical means of obtaining water supply is available.

Leave a Reply