Thermodynamics has its own unique vocabulary associated with it. A good understanding of the basic concepts forms a sound understanding of various topics discussed in thermodynamics preventing possible misunderstandings.
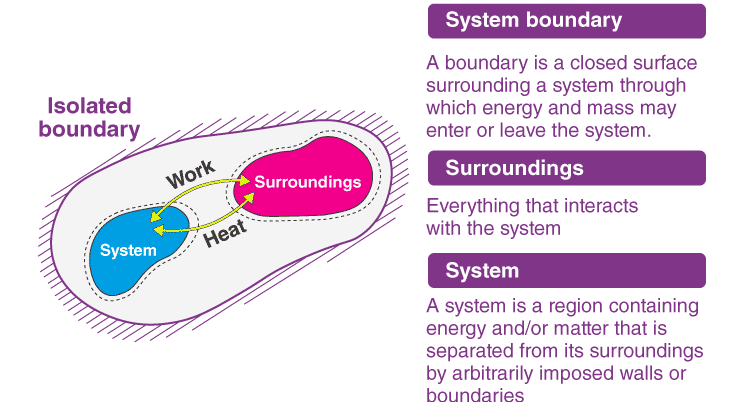
System
A thermodynamic system is a specific portion of matter with a definite boundary on which our attention is focused. The system boundary may be real or imaginary, fixed or deformable.
There are three types of systems:
- Isolated System – An isolated system cannot exchange energy and mass with its surroundings. The universe is considered an isolated system.
- Closed System – Across the boundary of the closed system, the transfer of energy takes place but the transfer of mass doesn’t take place. Refrigerator, compression of gas in the piston-cylinder assembly are examples of closed systems.
- Open System – In an open system, the mass and energy both may be transferred between the system and surroundings. A steam turbine is an example of an open system.
| Interactions of thermodynamic systems | |||
| Type of system | Mass flow | Work | Heat |
| Isolated System | ☓ | ☓ | ☓ |
| Open System | ✓ | ✓ | ✓ |
| Closed System | ☓ | ✓ | ✓ |
Surrounding
Everything outside the system that has a direct influence on the behaviour of the system is known as a surrounding.
Thermodynamic Process
A system undergoes a thermodynamic process when there is some energetic change within the system that is associated with changes in pressure, volume and internal energy.
There are four types of thermodynamic processes that have their unique properties, and they are:
- Adiabatic Process – A process where no heat transfer into or out of the system occurs.
- Isochoric Process – A process where no change in volume occurs and the system does no work.
- Isobaric Process – A process in which no change in pressure occurs.
- Isothermal Process – A process in which no change in temperature occurs.
Read More: Thermodynamic Process
A thermodynamic cycle is a process or a combination of processes conducted such that the initial and final states of the system are the same. A thermodynamic cycle is also known as cyclic operation or cyclic processes.
Thermodynamic Equilibrium
At a given state, all properties of a system have fixed values. Thus, if the value of even one property changes, the system’s state changes to a different one. In a system that is in equilibrium, no changes in the value of properties occur when it is isolated from its surroundings.
- When the temperature is the same throughout the entire system, we consider the system to be in thermal equilibrium.
- When there is no change in pressure at any point of the system, we consider the system to be in mechanical equilibrium.
- When the chemical composition of a system does not vary with time, we consider the system to be in chemical equilibrium.
- Phase equilibrium in a two-phase system is when the mass of each phase reaches an equilibrium level.
A thermodynamic system is said to be in thermodynamic equilibrium if it is in chemical equilibrium, mechanical equilibrium and thermal equilibrium and the relevant parameters cease to vary with time.
You may also want to check out these topics given below!
- Kelvin Planck Statement
- Darcy Weisbach Equation Derivation
- Kinetic Theory Of Gases Derivation
- Relation Between Kp And Kc

Leave a Reply