Category: 5. Thermodynamics
-

Perfect gases, gas laws, gas processes
The expansion and compression of gases, such as air and combustion gases, is a very important subject in the study of the operation of compressors, all types of reciprocating engines, gas turbines, and in pneumatic systems. We need to be able to predict how the volume, temperature and pressure of a gas inter-relate in a…
-
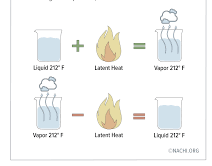
Latent heat
Further sensible heat energy will then be needed to raise its temperature to boiling point, followed by more latent heat (of vaporization) to change it into steam. See Figure 2.1.2. Figure 2.1.2 Sensible and latent heat Just as each substance has its own value of specific heat, so each substance has a value of latent heat of…
-

Specific heats of gases
Specific heats of gases Specific heat at constant pressure, cp. This is the quantity of heat energy supplied to raise 1 kg of the gas through 1°C or K, while the gas is at constant pressure. Think of 1 kg of gas trapped in a cylinder. As heat energy is added, the pressure will rise. If…
-

Power
It is not always useful to know only how much heat energy is needed to raise the temperature of a body. For instance, if you are boiling a kettle, you are more interested in how long it will be before you can make the tea. The quantity of heat energy needed has to be related…
-

Specific heat
The specific heat of a substance is the heat energy required to raise the temperature of unit mass of the substance by one degree. In terms of the quantities involved, the specific heat of a substance is the heat energy required to raise the temperature of 1 kg of the material by 1°C (or K, since they…
-
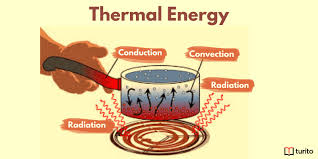
Heat energy
This chapter introduces heat energy by looking at the specific heat and latent heat of solids and gases. This provides the base knowledge required for many ordinary estimations of heat energy quantities in heating and cooling, such as are involved in many industrial processes, and in the production of steam from ice and water. The special…
-

Key Equations and Concepts
Ideal Gas Law: Internal Energy (U): Enthalpy (H): Entropy (S): Gibbs Free Energy (G):
-

Applications of Thermodynamics
Heat Engines: Refrigeration and Air Conditioning: Chemical Reactions: Phase Changes:
-
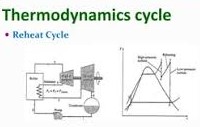
Thermodynamic Cycles:
Carnot Cycle: Idealized cycle consisting of two isothermal and two adiabatic processes.Otto Cycle: Used in gasoline engines, consisting of isentropic and isochoric processes.Rankine Cycle: Used in steam power plants, consisting of isentropic and isobaric processes.
-