Author: workhouse123
-
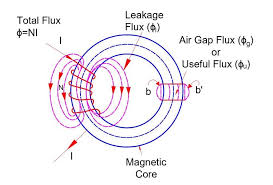
LEAKAGE FLUX
The magnetic flux that does not follow the intended path in a magnetic circuit is called leakage flux. Fig. 5.7 Magnetic circuit with leakage flux When some current is passed through a solenoid, as shown in Figure 5.7, magnetic flux is produced by it. Most of this flux is set up in the magnetic core and passes…
-

MAGNETIC CIRCUITS
The region around the magnet where its poles exhibit a force of attraction or repulsion is called magnetic field. The existence of the magnetic field at a point around the magnet can also be determined by placing a magnetic needle at that point, as shown in Figure 5.1. Although magnetic lines of force have no real existence and…
-

INTRODUCTION
The operation of all electrical machines such as DC machines, transformers, synchronous machines, and induction motors rely upon their magnetic circuits. The closed path followed by the magnetic lines of force is called a magnetic circuit. The operation of all the electrical devices (e.g., transformers, generators, motors, etc.) depends upon the magnetism produced by their…
-

SMALL NICKEL–CADMIUM CELLS
The various advantages of nickel–cadmium cells such as longer life, low maintenance cost, low internal resistance, etc. prompted the scientists to develop these cells in small sizes. Since the emf developed by these cells is 1.2 V, which is very near to the emf developed by dry cells (1.5 V), these cells have been developed…
-

COMPARISON BETWEEN LEAD–ACID AND NICKEL–IRON ALKALINE CELL
The two cells can be compared on the basis of the following particulars (Table 4.2): Table 4.2 Comparison of Lead-Acid and Nickel-Iron Cell 4.15 NICKEL–CADMIUM CELL It was developed by a Swedish scientist Waldemar Jungner in 1899. 4.15.1 Construction Cathode – Ni(OH)4 Anode – Cd (Cadmium) Electrolyte – KOH (Potassium hydroxide) of specific gravity 1.2. Its construction is similar to a nickel–iron cell with the…
-

NICKEL–IRON ALKALINE CELL
It is also known as Edison cell as it was developed by an American scientist Thomson A Edison in 1909. 4.13.1 Construction It contains two plates, that is, a positive plate (cathode) and a negative plate (anode). The active material of cathode is Ni(OH)4 and of anode is iron (Fe) when fully charged. The two plates are…
-

APPLICATIONS OF LEAD–ACID BATTERIES
Lead–acid batteries have innumerable commercial applications. Some of the important applications are as follows:
-

CARE AND MAINTENANCE OF LEAD–ACID BATTERIES
The average life of a lead–acid battery is two to four years depending upon its manufacturing qualities and technique. However, to obtain longer life and efficient service, the following points must be kept in view:
-

CHARGING OF LEAD–ACID BATTERY
Whenever terminal voltage of a battery falls below 1.8 V per cell, it is put under recharging. The following points must be kept in mind while charging a battery: Note: An over current may produce excessive heat and damage the battery. Generally, the following two methods are employed for charging a battery: Example 4.7 A battery…
-

EFFICIENCY OF A BATTERY
The efficiency of a battery (or cell) can be defined in the following two ways: Example 4.4 An alkaline cell is discharged at a steady current of 4 A for 12 hour, the average terminal voltage being 1.2 V to restore it to its original state of voltage, a steady current of 3 A for…