Author: workhouse123
-

CAPACITY OF A BATTERY
The quantity of electricity that a battery can delivery during single discharge until its terminal voltage falls to 1·8 V/cell is called the capacity of a battery. The capacity of a battery or cell is commercially expressed in ampere-hour and is generally denoted by A-H. Capacity of a battery or cell = IdTd ampere-hour Where Id =…
-

Working Principle of Lead–Acid Cell
When a lead–acid cell is ready for use, its positive plate is made of load peroxide (PbO2) chocolate brown in colour and the negative plate is of spongy lead (Pb) grey in colour. Both the plates are immersed in a dilute sulphuric acid (H2SO4) of specific gravity about 1.28. When a load is connected across…
-

GROUPING OF CELLS
A single cell can supply a very low current at low voltage. Generally, to operate electrical and electronic circuits, a large current at high voltage is required. Therefore, a number of cells are connected in series, parallel, and series–parallel grouping. 4.5.1 Series Grouping When a number of cells are connected in such a way that the…
-

IMPORTANT TERMS RELATING TO AN ELECTRIC CELL
The following are the important terms related to an electric cell: Fig. 4.1 Terminal voltage of a cell of no-lead and at load (a) Cell with internal resistance of no-load (b) Cell at load When load is applied, it delivers current I to the external (load) resistor R as shown in Figure 4.1(b). The potential difference across the terminals reduces to V volts…
-

IMPORTANT TERMS RELATING TO AN ELECTRIC CELL
The following are the important terms related to an electric cell: Fig. 4.1 Terminal voltage of a cell of no-lead and at load (a) Cell with internal resistance of no-load (b) Cell at load When load is applied, it delivers current I to the external (load) resistor R as shown in Figure 4.1(b). The potential difference across the terminals reduces to V volts…
-

ELECTRIC CELL
An electric device, such as battery, that converts chemical energy into electrical energy is known as electric cell. 4.2.1 Forming of a Cell An electric cell essentially consists of the following: When the two electrodes are immersed in a suitable electrolyte, different chemical actions take place on them and a potential difference is established between them.…
-

INTRODUCTION
Current not only flows through metallic conductors but also flows though some liquids called electrolytes.1 These liquids provide oppositely charged ions, and the conduction is due to the movement of these ions (not due to the movement of free electrons). The flow of current through electrolyte leads to chemical changes. Therefore, in this process, electrical energy…
-
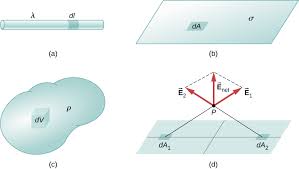
DIFFERENT WAYS OF CHARGE DISTRIBUTION
On a conductor or body, charge can be distributed in three ways: 3.11.1 Linear Charge Distribution When the charge is uniformly distributed over a line (straight or circular), the distribution is called linear charge distribution. If a charge Q is distributed over the length l of the conductor, then its linear charge density ( λ) is given by the relation: Hence,…
-
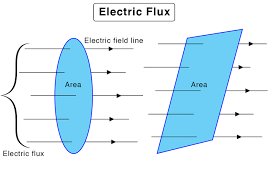
ELECTRIC FLUX THROUGH AN AREA
Electric flux through an area is defined as the number of electric lines of force passing perpendicularly through that area. It is a scalar quantity and is denoted by Ψ. Fig. 3.12 Electric flux through an area Consider an area S placed in an electric field and is a small area vector element, then small flux ( d Ψ ) passing through small area…
-

RELATION BETWEEN σ AND E
Consider a charge of + Q coulomb placed in a medium of relative permittivity εr, as shown in Figure 3.9. To determine electric flux density at point P, consider an imaginary sphere passing through point P having radius d. Fig. 3.9 Electric flux density at a point near a charged body The electric flux emanated by the charge in all directions = Q coulomb The surface…